The primary health care physician and the cancer patient: tips and strategies for managing sexual health
Cancer related sexual dysfunction
Worldwide, an estimated 14.1 million patients are diagnosed with cancer annually (1) and a majority of these individuals will become long-term cancer survivors (2). As greater numbers of survivors are living long after diagnosis and treatment, there is growing recognition that primary care physicians (PCPs) need to play an increasingly important role in addressing the numerous treatment-related side effects that impact quality of life for millions of cancer survivors (3). As PCPs are often in position to provide the majority of post-treatment medical care when survivors transition out of the oncology setting (4), they are, in a sense, on the “front lines” when it comes to managing long-term side effects for many survivors (5).
Sexual health is one of the most fundamental and long-lasting aspects of function that can be negatively affected by cancer treatment (6). Estimates of the prevalence of sexual dysfunction after cancer range from 40-100% (7-9), and affect both sexes. For both men and women, common problems includes disorders of sexual response (e.g., arousal, erectile dysfunction, ejaculatory dysfunction, reduced lubrication in females, chronic dyspareunia, orgasmic dysfunction etc.), and disorders of sexual desire and motivation (e.g., hypoactive sexual desire, reduced sexual motivation, body image disturbances, loss of sexual self-esteem etc.) (10). Without intervention, the detrimental impact of sexual dysfunction on cancer patients is significant and evidence suggests that these problems often get worse over time (6,11).
Because the sexual side effects of treatment are both profound and enduring, PCPs have a particularly important role to play in helping cancer survivors address and manage these problems. However, it has been shown that PCPs often do not feel comfortable managing sexual side effects (12) and that a lack of discussion with patients about sexual dysfunction has been associated with PCPs’ self-report of not receiving adequate preparation and/or formal training around survivorship care (13). Communication about sexual health is also hampered by a concrete lack of material resources such as clinical checklists, and educational materials (14,15). Thus it is imperative for PCPs to have a range of efficient strategies including language for communication, simple checklists for clinical inquiry and access to useful resources in order to facilitate communication with patients about sexual problems after cancer.
For the purpose of providing an overview of commonly reported cancer-related sexual health issues that PCPs can expect to encounter, we will begin by briefly describing frequently reported sexual symptoms and side effects related to common cancers. In other sections covered within this special journal issue, there is more in depth coverage of the specific sexual dysfunction that providers can expect to encounter with regard to particular diagnoses and treatments. Subsequently, we will address strategies for enhancing communication about sexual health between PCPs and survivors, offer tips for use of a model for clinical inquiry and clinical checklists, and make recommendations for resources to offer patients who are struggling with sexual problems after cancer. Finally, we will highlight clinical case examples that PCPs may encounter in their practice, and overview clinical next steps that a PCP might consider for the cases described.
Overview of common cancers and treatment-related sexual problems
Breast cancer
Treatment for breast cancer can involve surgery, chemotherapy, radiation, and/or hormone therapy. The experience of any one of these treatments is likely to impact the survivor’s sexual health (16). Therefore, research indicates that breast cancer survivors are significantly more likely to suffer from sexual problems when compared to the general population (17). Breast cancer survivors are likely to report libido changes, vaginal dysfunction (dryness, stenosis), different orgasm experiences, changes to body image, loss of intimacy, and a different relationship with their partner (16,18-20).
Gynecologic cancer
The treatment of gynecologic cancers is very likely to result in some form of sexual dysfunction (21). Survivors of gynecologic cancers experience worse sexual problems than women in the general population (22), regardless of specific site of their gynecologic cancer (e.g., endometrial, vulvar, cervical), treatment type, time from diagnosis and age (23,24). Their sexual dysfunction tends not to improve over time, and can intensify in nature (25). Surgery is a common component of treatment for gynecologic cancers and can result in long-term sexual health issues of pain, loss of sensation, changes in body image, vaginal dryness, difficulty reaching orgasm (26), and can trigger premature menopause symptoms including dyspareunia, and low libido (27). In addition, these women report that their treatments can alter their feelings of femininity, mood, self-esteem, and the way they relate to and discuss sexual issues with their partners (28-33). The addition of radiation and/or chemotherapy to the treatment regimen puts the patient at an increased risk of developing more severe sexual problems (34).
Prostate cancer
Because of the direct effect of treatment on sexual organs, men surviving prostate cancer commonly experience a significant impact on their sexual function as a result of treatment (9,35,36). Depending on a number of disease specific characteristics, prostate cancer patients may undergo a range of treatments including surgery, radiation, and hormonal therapy. Post-treatment, prostate cancer survivors commonly report symptoms of sexual dysfunction including erectile dysfunction, dry orgasms, urinary incontinence during orgasm, decreased satisfaction with orgasm, decreased penile length, body feminization and avoidance of sexual activities (37-54). As all active treatment options for prostate cancer are associated with compromised sexual functioning (55), the implications that these issues have on quality of life for prostate cancer survivors is critical for a provider to consider (36,55-59). In addition, prostate cancer survivors commonly endorse sexual bother even when function recovers, indicating that significant struggles remain in their efforts to cope with their functional decrements (60,61).
Testicular cancer
Sexual problems commonly reported by testicular cancer survivors include difficulties with sexual desire, ejaculatory difficulties and erectile dysfunction (62-65). These may be related to structural and emotional (body image) issues related orchiectomy, as well as retrograde ejaculation due to pelvic lymph node dissection. Following treatment these men often report reductions to sexual activity levels, and general sexual dissatisfaction (66,67).
Bladder cancer
Treatment for bladder cancer includes surgery, radiation, immunotherapy, and chemotherapy, with surgery being the most common course of action. For men, standard radical cystectomy is often associated with the loss of sexual function, most notably erectile dysfunction (68). For women, radical cystectomy is likely to create prominent sexual dysfunction including reduced libido, dyspareunia, decreased lubrication, and diminished ability or inability to achieve orgasm (69,70). In addition, men and women also suffer from the psychological impact of treatment in addition to their physical challenges. They often report body image concerns after urinary diversion following radical cystectomy (71-74), which is associated with significant loss of sexual function and satisfaction up to five years later (75).
Colorectal cancer
Surgery for colorectal cancer often causes nerve damage, and can cause erectile and ejaculatory dysfunction in men and desire, pain, and orgasm difficulties in women (76-82). Colorectal cancer survivors can report that their sexual function is affected by emotional reactions and adjustment to colostomy and their stoma, with notable concerns related to odor, flatulence, and diarrhea.
Head and neck cancer
Treatment for head and neck cancers can cause facial alterations/disfigurement, as well as persistent changes to saliva quality and/or quantity, breathing and speech (83). As the function and appearance of the head and neck region plays such a critical role in our social interactions (84), treatment can have implications for relationship and sexual function in survivors (85,86). Consequently, head and neck cancer survivors report feeling less attractive, reduced libido, and decreased satisfaction with their sexual relationships (87-91).
Hematologic malignancies
For those diagnosed with a hematologic malignancy, chemotherapy, total body irradiation, stem cell transplantation, and even the placement of a central venous catheter can significantly impact the patients’ body image, intimacy, and sexuality (92-95). Survivors can report erectile dysfunction in men, vaginal dryness in women, and pain and difficulty with orgasm for both men and women (94,96-98).
Childhood cancer
Those diagnosed with cancer at a young age are exposed to treatments that can impact their sexual health during critical developmental periods. Physically, treatment can impair their hormonal, vascular, genitourinary and neurological function, placing these survivors at risk for both sexual dysfunction (99-104), and infertility (105,106). Further, evidence suggests that even when young adult survivors of pediatric cancer report generally good health, they still have increased prevalence of sexual dysfunction (104). Psychosocially, they tend to be less sociable and more isolated, are less likely to marry, show greater restriction in their sexual behavior (e.g., masturbation, talking to friends about sex), delays in reaching sexual milestones (e.g., dating, intercourse), and decreased sexual interest and satisfaction with sex (107-113).
Communication about sexual dysfunction
A consistent theme across the literature has been that cancer patients and survivors rarely discuss issues of sexual function with their medical providers (114). As the patient begins their cancer treatment, the main focus for both the patient and oncology provider is on ensuring their survival. Therefore, it is not surprising that conversations with oncology providers about the short and long-term sexual health consequences of treatment either do not take place or are not well remembered (115,116). An estimated 0-37% of cancer survivors report that they had a discussion about sexual health with any member of their medical team (117-119). Similarly, oncology professionals report that they do not often discuss issues related to sexual health with their patients. In a survey of gynecologic oncologists, less than half reported that they took a patient sexual history at least 50% of the time (120). Similarly, providers treating women with ovarian cancer indicated that the overwhelming majority did not discuss sexual issues, despite acknowledging that these patients were likely to experience some form of sexual dysfunction following treatment (121). Like their colleagues in oncology, PCPs are also unlikely to discuss sexual health issues with cancer survivors who have completed active treatment. In a survey of primary health care physicians, over 60% of providers reported that they “never” or “rarely” addressed sexual dysfunction issues, and more than half were unlikely to initiate a conversation about sexual dysfunction with cancer survivors (122). Even when discussions occur with cancer survivors, they are often limited to the discussion of functional status, and rarely do issues related to the impact of sexual dysfunction on mood, quality of life, relationship functioning etc. get discussed (123,124).
This lack of discussion about sexual health stands in direct contrast to what medical professionals report regarding the importance they place on such issues, and their capability of delivering this care. For instance, nearly all gynecologic oncologists surveyed in one study reported that they were comfortable with taking the sexual history of their patients (120), and that issues related to sexual health consequences of cancer treatment should be discussed with patients (125).
Despite the lack of communication about sexual health after cancer, cancer patients/survivors report a consistent desire to have open dialogue with their medical providers about sexual issues (117,126). They are interested in conversations ranging from physiological changes that result from treatment, to the safety of sexual activity to psychological issues such as reassurances that their sexual issues are commonplace. Perhaps more importantly, cancer patients/survivors indicate that they are amenable to discussing how to resolve the impact of the changes to their sexual function and intimate relationships following cancer (116,117,123).
Challenges in sexual health communication
Despite the medical provider’s awareness of the importance of discussing sexual function, and the patient’s interest in receiving further information about sexual health issues, assessment and counseling about sex is not commonly a part of routine medical care across the world (114,127). There are numerous barriers to this important conversation on both sides of the examination table, and often neither the patient nor the PCP feels comfortable initiating conversations about sexual health.
From a patient’s perspective, he or she may experience challenges due to the patient/provider relationship, and a lack of accurate knowledge about sexual function and cancer. Moreover, patients report that if his or her provider does not bring up a medical issue, then it must not be of significant concern. Therefore patients are cautious about bringing up sexual dysfunction concerns because they are uncertain about its validity if their provider does not initiate (123,124,128) and they may be worried about feeling disrespected in such an interaction (128,129). During the particularly stressful period of time soon after a cancer diagnosis, patients are often overwhelmed with information and treatment planning and simply do not have the capacity to consider the sexual health implications of their cancer treatment (128,130). In addition, cancer patients may possess inaccurate beliefs about sexuality that reduce the likelihood that they will raise such issues with medical providers. For example, they may worry that cancer is contagious and can be spread through sexual acts, that sexual activity may impact their cancer recovery, or that side effects from cancer treatment make sexual activity impossible (131,132).
For the PCP, the literature points to types of barriers that bar effective communication about sexual health: patient characteristics, provider characteristics, and systems-based challenges. First, there are a number of patient characteristics that can discourage a medical provider from discussing sexual health; for example, age, gender, race/ethnicity, sexual orientation and partner status can all impact the provider’s initiative (121-123,133). In addition, the patient’s health prognosis, particularly in a palliative care setting, is likely to play a role in impacting whether the medical professional believes that the patient is interested in having a conversation about sexual health (123,124). Second, provider characteristics including their training background, knowledge about sexual health issues, and attitudes towards sex can negatively impact the likelihood of a conversation about the topic. Some medical providers recognize that they lack the experience and/or knowledge about sexual health issues that would allow them to feel confident with discussing it with their patients (121,122,133). Furthermore, medical providers report that it is frequently unclear as to which member of the multi-disciplinary medical team is responsible for initiating the conversation (121,123). Given that sex is often considered a taboo subject in many cultures not to be discussed openly, medical providers admit that they are sometimes embarrassed to openly speak about sexual issues, and consequently, avoid such intimate conversations (121,123). Finally, the medical system itself can make conversations about sexual health issues challenging. Physicians often have overloaded patient schedules and do not have sufficient time to thoroughly explore sexual functioning with each and every patient (116,120). Even when patients and providers discuss sexual dysfunction, there may be systemic difficulties surrounding the lack of resources available for the patient, and whether their health insurance would provide coverage for these issues (116,132). Given that cancer survivors may have complicated medical histories and a variety of other late effects of treatment, barriers such as time constraints and lack of experience and/or knowledge may be even more problematic for the PCP.
Strategies to address sexual health in a primary care setting
We suggest that the Five A’s Framework, a counseling model built on five basic components (ask, advise, assess, assist, and arrange), can provide an efficient and flexible structure for helping PCPs address sexual function with their patients (13,134). The first A (ask) underscores the PCP’s primary role in ensuring that patients know that sexual dysfunction is a medical issue that is commonly experienced by cancer survivors, and that this is a topic area which will be discussed during the course of their medical visit. We recommend that conversations about sexual dysfunction should be considered part of the routine review of systems. PCPs should aim to inquire about sexual function in an open-ended fashion. Common non-judgmental questions to begin initial inquiry and offer validation of the problem may include:
- “Many patients that I see express concerns about how their (treatment, disease) has affected sexual function. How has this been for you?”
- “Do you have concerns or worries about how your intimate relationship has been affected by your cancer treatment? Is this something you would like to talk about?”
- “In my experience, many people find that the kind of treatment you received can affect sexuality or intimacy. Do you have any questions for me about your experience?”
Closely related to providing validation is the PCP’s responsibility to let the patient know that he or she is willing to advise the patient as needed. That is, this initial communication also conveys an important implicit message that treatment for sexual problems after cancer is available. The next step involves the need to adequately assess the problem in a manner which is efficient and also allows the PCP to identify next steps for intervention. Initially, PCPs can consider the use of paper and pencil screening tools as part of their regular intake paperwork. This serves the purpose of briefly assessing sexual function in cancer survivors evaluating which patients may require further evaluation. The use of such a screening measure can help to address barriers such as providers who have limited time, feel uncomfortable screening certain patients (e.g., an older patient who is recently widowed) or feel embarrassed by bringing up sexuality directly with patients. Providers can swiftly review patient’s responses and utilize endorsed items as a starting point for a more thorough conversation about sexual dysfunction. Even for providers who currently feel comfortable having a discussion with patients about sexuality, screening tools may serve as a guide to review symptoms that might be present post-cancer treatment.
PCPs can consider several easily accessible and widely utilized instruments for the evaluation of sexual dysfunction. For female patients, the Female Sexual Function Index (FSFI) (135) is a commonly used 19-item self-report measure originally developed to assess female sexual function in women of any age, including pre- and post-menopause, in the general population and takes approximately 15 minutes to complete. The scale assesses function over the past month in several domains: desire, arousal, lubrication, orgasm, satisfaction, and pain (136) and has been utilized and validated in cancer patients and survivors (137,138). For providers who are particularly conscious of patient burden, they can consider the abbreviated 6-item version of the FSFI, though this is not recommended as it does not provide as much clinical information as the full scale (139). Those seeking further information about this scale can find additional resources at www.FSFIquestionnaire.com (135). In male patients, providers can consider the International Index of Erectile Function (IIEF) (140). The scale is a 15-item self-report measure developed to assess erectile function in men in the general population and has been utilized in studies with cancer patients and survivors, particularly with prostate cancer populations (137). The IIEF measures function over the past month in the following domains: erectile function, orgasm, desire, intercourse satisfaction, and overall satisfaction. As with the FSFI, there are also briefer versions of the IIEF which may be considered for use as a screening tool (137,141). Though potentially useful for the PCP, the IIEF is limited as it primarily evaluates for erectile dysfunction, and other sexual dysfunctions in men could be overlooked. Providers should be aware that there are a large number of other sexual function measures available. Other screening checklists of sexual dysfunction in men have been developed, and are of value for the PCP. However, providers should exercise clinical judgment as these measures may not have been empirically validated in cancer patients. For example, a brief general screening tool (versions for both men and women) has recently been provided by Hatzichristou et al. (142) and can serve as a guide for PCPs looking to incorporate such a checklist with their patients. When making a decision as to which measure to utilize, the validity and reliability of the measure, as well as the time it takes to complete and the breath of the assessment are crucial factors for the PCP to consider.
After the PCP has clarified the problem, patients then need to be assisted by receiving necessary resources such as information sheets, and access to educational books and websites or potentially PCPs may need to arrange further intervention other providers. It is possible that patients may need to see a specialist such as mental health professional, a sexual health counselor, and an urologist specializing in sexual medicine or a menopause specialist. It is also our belief that the final step of making arrangements for additional evaluation with specialists also includes arranging to follow-up with patients at subsequent visits. Such follow-up communicates the message that the PCP takes these issues seriously, is willing to communicate about sexual health, and reassures the patient that remaining challenges can be addressed. The following section contains specific recommendations and tips that PCPs can use in the context of assisting and arranging care for patients.
It is important to consider that the optimal delivery of survivorship care, including attention to sexual health, may require more time than the PCP has with his or her patient. Other comparable models for intervention, such as the ALLOW algorithm (143) (ask, legitimize, limitations, open up for further discussion/evaluation, work together to develop a treatment plan) encourages the PCP to discuss sexual health issues, and help the patient find adequate resolution for their identified dysfunction. Similar to the five A’s Framework, this model also acknowledges that whatever the concrete constraints of clinical practice, the key elements for interaction revolve around inquiry, validation and provision of resources as needed.
Just as it is important for PCPs to have access to basic information about managing common sexual side effects, it is equally important that they have access to patient education and self-help resources which can be made available to both male and female patients. First, one should identify local professionals capable of providing more specialized treatment for sexual health issues. These professionals could include members of the following disciplines:
- Urologist/sexual medicine specialist/uro-gynecologist;
- Gynecologist/menopause specialist;
- Endocrinologist;
- Clinical psychologist/sexual health counselor;
- Pelvic floor physical therapist.
Often it is helpful to build a resource list of local specialists which may be cultivated through a number of professional societies that specialize in sexual medicine/sexual therapy. Many of these societies not only have listings to find providers but also have excellent educational material related to sexuality after cancer. Examples of such societies that a PCP can consider include:
- International Society for Sexual Medicine (http://www.issm.info);
- North American Menopause Society (http://www.menopause.org);
- International Society for Sexuality and Cancer (http://www.issc.nu);
- Women’s Health and Research Institute of Australia (http://www.whria.com.au);
- American Association of Sexuality Educators, Counselors and Therapists (http://www.aasect.org).
There are also a wide range of websites that are aimed at patient education and yield a significant amount of high quality information. Examples of patient websites include:
- American Cancer Society: Sexuality for the Woman with Cancer: http://www.cancer.org/treatment/treatmentsandsideeffects/physicalsideeffects/sexualsideeffectsinwomen/sexualityforthewoman/sexuality-for-the-woman-with-cancer-toc;
- American Cancer Society: Sexuality for the Man with Cancer: http://www.cancer.org/treatment/treatmentsandsideeffects/physicalsideeffects/sexualsideeffectsinmen/sexualityfortheman/sexuality-for-the-man-with-cancer-toc;
- Macmillan Cancer Support: Effects of Cancer on Sexuality: http://www.macmillan.org.uk/Cancerinformation/Livingwithandaftercancer/Relationshipscommunication/Sexuality/Effectofcanceronsexuality.aspx.
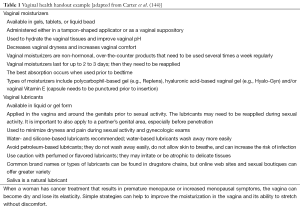
Acknowledging that PCPs often work under multiple constraints, including the need to address a very wide range of issues in a very compressed amount of time, it is ideal if primary care practices can have resource or “tip” sheets for cancer survivors that overview common problems such as vaginal dryness or lack of libido. Such resource sheets can save time and be an enormous resource for patients. It is our recommendation to work with either a nurse in the primary care practice or a local partner (pelvic floor therapist, sex therapist) to create a resource sheet. An excellent example is the suggested patient handout created by Carter et al. [2011] (144) for women to promote vaginal health after cancer (Table 1).
Clinical case examples
Case example 1—Mariel, a 28-year-old non-Hodgkin lymphoma survivor
Medical history
Mariel was diagnosed with diffuse large cell non-Hodgkin lymphoma at the age of 14 after presenting with a neck mass. She experienced a long course of treatment which included focal radiation and multi-agent chemotherapy. Unfortunately, Mariel relapsed at age 16, and required a stem-cell transplant with additional chemotherapy and total body radiation. Thus, Mariel was in and out of cancer treatment for most of her high school years.
Sexual health challenges
While in college, Mariel began noticing that she was behind her peers in terms of her social development. Despite being an excellent student academically, Mariel started to become aware that her friends were choosing to spend more time with their boyfriends, rather than with her. She wants to start dating more regularly, but does not feel comfortable in this domain. She has sexual desire, but has felt awkward during previous sexual interactions. Mariel says: “I was not really depressed or anything during my treatment, I was a pretty happy person no matter how bad things felt. When I got sick and left school everyone was starting to date, and when I got better and came back it was like everyone was just having sex—I couldn’t figure out what I missed and didn’t know what I was supposed to do about even talking to a guy without feeling uncomfortable. Socially it has been a total disaster. I went from being in a protective bubble to being dropped in a huge college campus that was like one big party. I feel so out of place sometimes.”
Considerations for the primary health care physician
This patient faces issues that are common among childhood cancer survivors. While cancer treatment may have untoward effects on physical function that can lead to sexual problems, this case highlights the psychosocial late effects for this vulnerable population. Despite coping very well with the immediate stressors of her cancer treatment, Mariel’s social and psychological development was dramatically interrupted during her adolescence. Losing out on opportunities to be a “normal” teenager can make sexual milestones (e.g., dating, physical intimacy etc.) seem more intimidating especially if the survivor is aware that he/she is “behind”. Despite presentation of overall robust health, the PCP needs to be aware that childhood cancer treatment often results in psychosexual developmental delays. Especially because young adults may not be comfortable initiating this conversation, the PCP’s inquiry signifies an important first step to helping young adult survivors begin to readjust their expectations and return to normative function. The PCP can play an important role in encouraging that the childhood cancer survivor seek active psychotherapy, or to consider connecting with other childhood cancer survivors both in-person and via the internet in order to normalize their experiences.
Case example 2—Carson, a 62-year-old prostate cancer survivor
Medical history
Carson is a 62-year-old semi-retired, married attorney who was diagnosed 18 months earlier with high-risk prostate cancer. He underwent nerve-sparing radical prostatectomy, external beam radiation and was placed on androgen-deprivation therapy for 18 months. Carson has been on this therapy for 6 months now.
Sexual health challenges
Carson initially presented to his PCP for consultation about difficulty with his sleep. Secondary to this, he casually reported to his PCP that he had been experiencing low mood, lack of energy and erectile dysfunction for several months now. Upon further inquiry, Carson recalled that although desire is diminished, it is not absent and that his lack of sexual function is primarily related to impaired erectile function. He is otherwise healthy and is not taking any medications. He reports to be happily married with a supportive wife.
Considerations for the primary health care physician
Often, patients will present to their PCP with health-related concerns that are not directly tied to their sexual health. As discussed previously, patients are often reticent to speak about their sexual dysfunction for a variety of reasons. It will be important for a PCP to be mindful of potential opportunities for further evaluation related to sexual health concerns. In Carson’s case, erectile dysfunction may be a side effect of his surgery and/or radiation therapy (due to local damage to the nerves and/or blood vessels). While this may be long-lasting for some patients, many can recover their erectile function to a certain extent. Androgen deprivation may further result in erectile dysfunction due to depletion of testosterone. In addition, anxiety and depression may also play a role. Carson’s case presents an excellent opportunity for the PCP to collaborate with a multi-disciplinary team in order to help Carson effectively manage his dysfunction. In addition to communicating with his oncologist, it may be helpful to connect with a urologist who specializes in men’s sexual health, and a clinical psychologist who can help him and his wife navigate his adjustment to post-cancer sexual functioning.
Case example 3—Mackenzie, a 47-year-old breast cancer survivor
Medical history
Mackenzie is a 47-year-old female breast cancer survivor who has come for her yearly physical. She was diagnosed with ER + PR + HER2 breast cancer at age 45 and was treated with unilateral mastectomy, radiation therapy and chemotherapy including doxorubicin, cyclophosphamide and trastuzumab. She is currently taking tamoxifen. Mackenzie stopped menstruating during her active therapy and has since not resumed her menses.
Sexual health challenges
Mackenzie has been married for 15 years, and has two school-age children with her husband. She works full-time as a pharmacist, and often comes home tired after a long day at work. She notes that sex is very painful despite copious use of lubricants. Because of this discomfort, she has stopped having regular sexual activity with her husband. She is concerned about the pain and remarks that since ending her treatment the situation with her sex life has not improved. As a result of the cessation of regular sexual activity, she has noticed a decline in the quality of her marital relationship.
Considerations for the primary health care physician
Sexual health concerns can often be the tip of the iceberg for a PCP, as sexual dysfunction can be associated with work-life distress, as well as marital dysfunction. Strictly from a sexual health perspective, Mackenzie is likely suffering from pain during intercourse due to vaginal dryness and vaginal atrophy. This is likely related to cessation of menses/early menopause (typically with cyclophosphamide) leading to loss of lubrication and/or as a side effect of the ongoing treatment with tamoxifen. Consequently, addressing this issue through the use of non-hormonal moisturizers, local topical estrogen use, and/or vaginal dilators with the assistance of a gynecologist and/or pelvic floor physical therapists could be beneficial for the patient. Also important for Mackenzie are the relationship sequelae of this dysfunction. The PCP is in an ideal situation to recommend that Mackenzie explore marital therapy in order to address the discord that resulted from her sexual dysfunction, and to help her understand that relationship function and sexual satisfaction are highly correlated.
Conclusions
Parallel to rapid advances in care, the number of long-term cancer survivors worldwide continues to grow at a significant pace. Consequently, these survivors bring a range of long-term side effects to their PCPs that need to address. As data demonstrate, PCPs want to provide excellent survivorship care yet often feel unprepared to do so. It is imperative that PCPs received straightforward recommendations and strategies to help their patients. Fortunately, the issue of sexuality after cancer is garnering greater attention and there are now more resources available for both professionals and patients than ever before. It is our hope that when PCPs appreciate the multifactorial etiologies for sexual symptoms in cancer survivors, have straightforward language and confidence to inquire about such symptoms, and have the tools to address and manage them and/or have readily available resources to call upon for additional evaluation, that they will feel more encouraged and empowered to not only assess patient function as it relates to sexuality post-cancer, but to also directly address sexual problems with patients as they are presented.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- UK CR. Worldwide cancer statistics 2012. Available online: http://www.cancerresearchuk.org/cancer-info/cancerstats/world/cancer-worldwide-the-global-picture
- DeSantis CE, Lin CC, Mariotto AB, et al. Cancer treatment and survivorship statistics, 2014. CA Cancer J Clin 2014;64:252-71. [PubMed]
- Grunfeld E. Primary care physicians and oncologists are players on the same team. J Clin Oncol 2008;26:2246-7. [PubMed]
- Grant M, Economou D, Ferrell B, et al. Educating health care professionals to provide institutional changes in cancer survivorship care. J Cancer Educ 2012;27:226-32. [PubMed]
- Nekhlyudov L, Wenger N. Institute of medicine recommendations for improving the quality of cancer care: what do they mean for the general internist? J Gen Intern Med 2014;29:1404-9. [PubMed]
- Darwish-Yassine M, Berenji M, Wing D, et al. Evaluating long-term patient-centered outcomes following prostate cancer treatment: findings from the Michigan Prostate Cancer Survivor study. J Cancer Surviv 2014;8:121-30. [PubMed]
- Derogatis LR, Kourlesis SM. An approach to evaluation of sexual problems in the cancer patient. CA Cancer J Clin 1981;31:46-50. [PubMed]
- Ganz PA, Rowland JH, Desmond K, et al. Life after breast cancer: understanding women’s health-related quality of life and sexual functioning. J Clin Oncol 1998;16:501-14. [PubMed]
- Stanford JL, Feng Z, Hamilton AS, et al. Urinary and sexual function after radical prostatectomy for clinically localized prostate cancer: the Prostate Cancer Outcomes Study. JAMA 2000;283:354-60. [PubMed]
- Basson R, Schultz WW. Sexual sequelae of general medical disorders. Lancet 2007;369:409-24. [PubMed]
- Bakewell RT, Volker DL. Sexual dysfunction related to the treatment of young women with breast cancer. Clin J Oncol Nurs 2005;9:697-702. [PubMed]
- Skolarus TA, Holmes-Rovner M, Northouse LL, et al. Primary care perspectives on prostate cancer survivorship: implications for improving quality of care. Urol Oncol 2013;31:727-32. [PubMed]
- Park ER, Bober SL, Campbell EG, et al. General internist communication about sexual function with cancer survivors. J Gen Intern Med 2009;24:S407-11. [PubMed]
- Tsai YF. Nurses’ facilitators and barriers for taking a sexual history in Taiwan. Appl Nurs Res 2004;17:257-64. [PubMed]
- Ussher JM, Perz J, Gilbert E, et al. Talking about sex after cancer: a discourse analytic study of health care professional accounts of sexual communication with patients. Psychol Health 2013;28:1370-90. [PubMed]
- Katz A. Breast cancer and women’s sexuality. Am J Nurs 2011;111:63-7. [PubMed]
- Brédart A, Dolbeault S, Savignoni A, et al. Prevalence and associated factors of sexual problems after early-stage breast cancer treatment: results of a French exploratory survey. Psychooncology 2011;20:841-50. [PubMed]
- Dizon DS. Quality of life after breast cancer: survivorship and sexuality. Breast J 2009;15:500-4. [PubMed]
- Gilbert E, Ussher JM, Perz J. Sexuality after breast cancer: a review. Maturitas 2010;66:397-407. [PubMed]
- Elmir R, Jackson D, Beale B, et al. Against all odds: Australian women’s experiences of recovery from breast cancer. J Clin Nurs 2010;19:2531-8. [PubMed]
- Abbott-Anderson K, Kwekkeboom KL. A systematic review of sexual concerns reported by gynecological cancer survivors. Gynecol Oncol 2012;124:477-89. [PubMed]
- Lindau ST, Gavrilova N, Anderson D. Sexual morbidity in very long term survivors of vaginal and cervical cancer: a comparison to national norms. Gynecol Oncol 2007;106:413-8. [PubMed]
- Golbasi Z, Erenel AS. The quality of sexual life in women with gynaecological cancers. Arch Gynecol Obstet 2012;285:1713-7. [PubMed]
- Wilmoth MC, Hatmaker-Flanigan E, LaLoggia V, et al. Ovarian cancer survivors: qualitative analysis of the symptom of sexuality. Oncol Nurs Forum 2011;38:699-708. [PubMed]
- Rodrigues AC, Teixeira R, Teixeira T, et al. Impact of pelvic radiotherapy on female sexuality. Arch Gynecol Obstet 2012;285:505-14. [PubMed]
- Carter J, Stabile C, Gunn A, et al. The physical consequences of gynecologic cancer surgery and their impact on sexual, emotional, and quality of life issues. J Sex Med 2013;10:21-34. [PubMed]
- Parker WH, Jacoby V, Shoupe D, et al. Effect of bilateral oophorectomy on women’s long-term health. Womens Health (Lond Engl) 2009;5:565-76. [PubMed]
- Akyüz A, Güvenç G, Ustünsöz A, et al. Living with gynecologic cancer: experience of women and their partners. J Nurs Scholarsh 2008;40:241-7. [PubMed]
- Cleary V, Hegarty J. Understanding sexuality in women with gynaecological cancer. Eur J Oncol Nurs 2011;15:38-45. [PubMed]
- Gilbert E, Ussher JM, Perz J. Sexuality after gynaecological cancer: a review of the material, intrapsychic, and discursive aspects of treatment on women’s sexual-wellbeing. Maturitas 2011;70:42-57. [PubMed]
- Afiyanti Y, Milanti A. Physical sexual and intimate relationship concerns among Indonesian cervical cancer survivors: A phenomenological study. Nurs Health Sci 2012. [Epub ahead of print]. [PubMed]
- Burns M, Costello J, Ryan-Woolley B, et al. Assessing the impact of late treatment effects in cervical cancer: an exploratory study of women’s sexuality. Eur J Cancer Care (Engl) 2007;16:364-72. [PubMed]
- Greenwald HP, McCorkle R. Sexuality and sexual function in long-term survivors of cervical cancer. J Womens Health (Larchmt) 2008;17:955-63. [PubMed]
- Eifel PJ, Berek JS, Markman MA. Cancer of the cervix, vagina, and vulva. In: DeVita VT Jr, Lawrence TS, Rosenberg SA. eds. Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, PA: Lippincott, Williams & Wilkins, 2011.
- Litwin MS, Hays RD, Fink A, et al. Quality-of-life outcomes in men treated for localized prostate cancer. JAMA 1995;273:129-35. [PubMed]
- Helgason AR, Adolfsson J, Dickman P, et al. Waning sexual function--the most important disease-specific distress for patients with prostate cancer. Br J Cancer 1996;73:1417-21. [PubMed]
- Penson DF, McLerran D, Feng Z, et al. 5-year urinary and sexual outcomes after radical prostatectomy: results from the prostate cancer outcomes study. J Urol 2005;173:1701-5. [PubMed]
- Choi JM, Nelson CJ, Stasi J, et al. Orgasm associated incontinence (climacturia) following radical pelvic surgery: rates of occurrence and predictors. J Urol 2007;177:2223-6. [PubMed]
- Guay A, Seftel AD. Sexual foreplay incontinence in men with erectile dysfunction after radical prostatectomy: a clinical observation. Int J Impot Res 2008;20:199-201. [PubMed]
- Nilsson AE, Carlsson S, Johansson E, et al. Orgasm-associated urinary incontinence and sexual life after radical prostatectomy. J Sex Med 2011;8:2632-9. [PubMed]
- Koeman M, van Driel MF, Schultz WC, et al. Orgasm after radical prostatectomy. Br J Urol 1996;77:861-4. [PubMed]
- Mitchell SA, Jain RK, Laze J, et al. Post-prostatectomy incontinence during sexual activity: a single center prevalence study. J Urol 2011;186:982-5. [PubMed]
- Tal R, Valenzuela R, Aviv N, et al. Persistent erectile dysfunction following radical prostatectomy: the association between nerve-sparing status and the prevalence and chronology of venous leak. J Sex Med 2009;6:2813-9. [PubMed]
- Dubbelman YD, Dohle GR, Schröder FH. Sexual function before and after radical retropubic prostatectomy: A systematic review of prognostic indicators for a successful outcome. Eur Urol 2006;50:711-8; discussion 718-20. [PubMed]
- Schover LR, Fouladi RT, Warneke CL, et al. Defining sexual outcomes after treatment for localized prostate carcinoma. Cancer 2002;95:1773-85. [PubMed]
- Fraiman MC, Lepor H, McCullough AR. Changes in Penile Morphometrics in Men with Erectile Dysfunction after Nerve-Sparing Radical Retropubic Prostatectomy. Mol Urol 1999;3:109-115. [PubMed]
- Gontero P, Galzerano M, Bartoletti R, et al. New insights into the pathogenesis of penile shortening after radical prostatectomy and the role of postoperative sexual function. J Urol 2007;178:602-7. [PubMed]
- Savoie M, Kim SS, Soloway MS. A prospective study measuring penile length in men treated with radical prostatectomy for prostate cancer. J Urol 2003;169:1462-4. [PubMed]
- Munding MD, Wessells HB, Dalkin BL. Pilot study of changes in stretched penile length 3 months after radical retropubic prostatectomy. Urology 2001;58:567-9. [PubMed]
- Lips IM, van Gils CH, van der Heide UA, et al. Health-related quality of life 3 years after high-dose intensity-modulated radiotherapy with gold fiducial marker-based position verification. BJU Int 2009;103:762-7. [PubMed]
- Sanda MG, Dunn RL, Michalski J, et al. Quality of life and satisfaction with outcome among prostate-cancer survivors. N Engl J Med 2008;358:1250-61. [PubMed]
- Potosky AL, Reeve BB, Clegg LX, et al. Quality of life following localized prostate cancer treated initially with androgen deprivation therapy or no therapy. J Natl Cancer Inst 2002;94:430-7. [PubMed]
- Gacci M, Simonato A, Masieri L, et al. Urinary and sexual outcomes in long-term (5+ years) prostate cancer disease free survivors after radical prostatectomy. Health Qual Life Outcomes 2009;7:94. [PubMed]
- Elliott S, Latini DM, Walker LM, et al. Androgen deprivation therapy for prostate cancer: recommendations to improve patient and partner quality of life. J Sex Med 2010;7:2996-3010. [PubMed]
- Parker WR, Montgomery JS, Wood DP Jr. Quality of life outcomes following treatment for localized prostate cancer: is there a clear winner? Curr Opin Urol 2009;19:303-8. [PubMed]
- Helgason AR, Fredrikson M, Adolfsson J, et al. Decreased sexual capacity after external radiation therapy for prostate cancer impairs quality of life. Int J Radiat Oncol Biol Phys 1995;32:33-9. [PubMed]
- Fergus KD, Gray RE, Fitch MI. Sexual dysfunction and the preservation of manhood: experiences of men with prostate cancer. J Health Psychol 2002;7:303-16. [PubMed]
- Penson DF, Feng Z, Kuniyuki A, et al. General quality of life 2 years following treatment for prostate cancer: what influences outcomes? Results from the prostate cancer outcomes study. J Clin Oncol 2003;21:1147-54. [PubMed]
- Zhou ES, Penedo FJ, Lewis JE, et al. Perceived stress mediates the effects of social support on health-related quality of life among men treated for localized prostate cancer. J Psychosom Res 2010;69:587-90. [PubMed]
- Parker WR, Wang R, He C, et al. Five year expanded prostate cancer index composite-based quality of life outcomes after prostatectomy for localized prostate cancer. BJU Int 2011;107:585-90. [PubMed]
- Benedict C, Traeger L, Dahn JR, et al. Sexual bother in men with advanced prostate cancer undergoing androgen deprivation therapy. J Sex Med 2014;11:2571-80. [PubMed]
- Brodsky MS. Testicular cancer survivors’ impressions of the impact of the disease on their lives. Qualitative Health Research 1995;5:78-96.
- Kuczyk M, Machtens S, Bokemeyer C, et al. Sexual function and fertility after treatment of testicular cancer. Curr Opin Urol 2000;10:473-7. [PubMed]
- Dahl AA, Bremnes R, Dahl O, et al. Is the sexual function compromised in long-term testicular cancer survivors? Eur Urol 2007;52:1438-47. [PubMed]
- Jonker-Pool G, Van de Wiel HB, Hoekstra HJ, et al. Sexual functioning after treatment for testicular cancer--review and meta-analysis of 36 empirical studies between 1975-2000. Arch Sex Behav 2001;30:55-74. [PubMed]
- Rossen P, Pedersen AF, Zachariae R, et al. Sexuality and body image in long-term survivors of testicular cancer. Eur J Cancer 2012;48:571-8. [PubMed]
- Nazareth I, Lewin J, King M. Sexual dysfunction after treatment for testicular cancer: a systematic review. J Psychosom Res 2001;51:735-43. [PubMed]
- Zippe CD, Raina R, Massanyi EZ, et al. Sexual function after male radical cystectomy in a sexually active population. Urology 2004;64:682-5. [PubMed]
- El-Bahnasawy MS, Osman Y, El-Hefnawy A, et al. Radical cystectomy and urinary diversion in women: impact on sexual function. Scand J Urol Nephrol 2011;45:332-8. [PubMed]
- Zippe CD, Raina R, Shah AD, et al. Female sexual dysfunction after radical cystectomy: a new outcome measure. Urology 2004;63:1153-7. [PubMed]
- Weijmar Schultz WC, Van De Wiel HB, Hahn DE, et al. Psychosexual functioning after treatment for gynecological cancer: an integrative model, review of determinant factors and clinical guidelines. Int J Gynecol Cancer 1992;2:281-90. [PubMed]
- Rosen RC, Taylor JF, Leiblum SR, et al. Prevalence of sexual dysfunction in women: results of a survey study of 329 women in an outpatient gynecological clinic. J Sex Marital Ther 1993;19:171-88. [PubMed]
- Bjerre BD, Johansen C, Steven K. A questionnaire study of sexological problems following urinary diversion in the female patient. Scand J Urol Nephrol 1997;31:155-60. [PubMed]
- Hart S, Skinner EC, Meyerowitz BE, et al. Quality of life after radical cystectomy for bladder cancer in patients with an ileal conduit, cutaneous or urethral kock pouch. J Urol 1999;162:77-81. [PubMed]
- Takenaka A, Hara I, Soga H, et al. Assessment of long-term quality of life in patients with orthotopic neobladder followed for more than 5 years. Int Urol Nephrol 2011;43:749-54. [PubMed]
- Thong MS, Mols F, Lemmens VE, et al. Impact of preoperative radiotherapy on general and disease-specific health status of rectal cancer survivors: a population-based study. Int J Radiat Oncol Biol Phys 2011;81:e49-58. [PubMed]
- Schmidt C, Daun A, Malchow B, et al. Sexual impairment and its effects on quality of life in patients with rectal cancer. Dtsch Arztebl Int 2010;107:123-30. [PubMed]
- Li CC. Sexuality among patients with a colostomy: an exploration of the influences of gender, sexual orientation, and Asian heritage. J Wound Ostomy Continence Nurs 2009;36:288-96. [PubMed]
- Larson DW, Davies MM, Dozois EJ, et al. Sexual function, body image, and quality of life after laparoscopic and open ileal pouch-anal anastomosis. Dis Colon Rectum 2008;51:392-6. [PubMed]
- Doeksen A, Gooszen JA, van Duijvendijk P, et al. Sexual and urinary functioning after rectal surgery: a prospective comparative study with a median follow-up of 8.5 years. Int J Colorectal Dis 2011;26:1549-57. [PubMed]
- Brændengen M, Tveit KM, Bruheim K, et al. Late patient-reported toxicity after preoperative radiotherapy or chemoradiotherapy in nonresectable rectal cancer: results from a randomized Phase III study. Int J Radiat Oncol Biol Phys 2011;81:1017-24. [PubMed]
- Traa MJ, De Vries J, Roukema JA, et al. Sexual (dys)function and the quality of sexual life in patients with colorectal cancer: a systematic review. Ann Oncol 2012;23:19-27. [PubMed]
- Bjordal K, de Graeff A, Fayers PM, et al. A 12 country field study of the EORTC QLQ-C30 (version 3.0) and the head and neck cancer specific module (EORTC QLQ-H&N35) in head and neck patients. EORTC Quality of Life Group. Eur J Cancer 2000;36:1796-807. [PubMed]
- Breitbart W, Holland J. Psychosocial aspects of head and neck cancer. Semin Oncol 1988;15:61-9. [PubMed]
- Chaukar DA, Walvekar RR, Das AK, et al. Quality of life in head and neck cancer survivors: a cross-sectional survey. Am J Otolaryngol 2009;30:176-80. [PubMed]
- Funk GF, Karnell LH, Christensen AJ. Long-term health-related quality of life in survivors of head and neck cancer. Arch Otolaryngol Head Neck Surg 2012;138:123-33. [PubMed]
- Gamba A, Romano M, Grosso IM, et al. Psychosocial adjustment of patients surgically treated for head and neck cancer. Head Neck 1992;14:218-23. [PubMed]
- Gritz ER, Carmack CL, de Moor C, et al. First year after head and neck cancer: quality of life. J Clin Oncol 1999;17:352-60. [PubMed]
- Singer S, Danker H, Dietz A, et al. Sexual problems after total or partial laryngectomy. Laryngoscope 2008;118:2218-24. [PubMed]
- Meyers AD, Aarons B, Suzuki B, et al. Sexual behavior following laryngectomy. Ear Nose Throat J 1980;59:327-9. [PubMed]
- Moreno KF, Khabbaz E, Gaitonde K, et al. Sexuality after treatment of head and neck cancer: findings based on modification of sexual adjustment questionnaire. Laryngoscope 2012;122:1526-31. [PubMed]
- Chatterjee R, Kottaridis PD, McGarrigle HH, et al. Management of erectile dysfunction by combination therapy with testosterone and sildenafil in recipients of high-dose therapy for haematological malignancies. Bone Marrow Transplant 2002;29:607-10. [PubMed]
- Yi JC, Syrjala KL. Sexuality after hematopoietic stem cell transplantation. Cancer J 2009;15:57-64. [PubMed]
- Milroy CL, Jones KP. Gynecologic care in hematopoietic stem cell transplant patients: a review. Obstet Gynecol Surv 2010;65:668-79. [PubMed]
- Møller T, Adamsen L. Hematologic patients’ clinical and psychosocial experiences with implanted long-term central venous catheter: self-management versus professionally controlled care. Cancer Nurs 2010;33:426-35. [PubMed]
- Humphreys CT, Tallman B, Altmaier EM, et al. Sexual functioning in patients undergoing bone marrow transplantation: a longitudinal study. Bone Marrow Transplant 2007;39:491-6. [PubMed]
- Liptrott SJ, Shash E, Martinelli G. Sexuality in patients undergoing haematopoietic stem cell transplantation. Int J Hematol 2011;94:519-24. [PubMed]
- Slovacek L, Slanska I, Slovackova B, et al. Sexual dysfunction in females treated with peripheral blood progenitor cell transplantation: review of literature. Bratisl Lek Listy 2010;111:457-60. [PubMed]
- Schover LR. Sexuality and fertility after cancer. Hematology Am Soc Hematol Educ Program 2005.523-7. [PubMed]
- Tromp K, Claessens JJ, Knijnenburg SL, et al. Reproductive status in adult male long-term survivors of childhood cancer. Hum Reprod 2011;26:1775-83. [PubMed]
- Green DM, Kawashima T, Stovall M, et al. Fertility of female survivors of childhood cancer: a report from the childhood cancer survivor study. J Clin Oncol 2009;27:2677-85. [PubMed]
- Green DM, Kawashima T, Stovall M, et al. Fertility of male survivors of childhood cancer: a report from the Childhood Cancer Survivor Study. J Clin Oncol 2010;28:332-9. [PubMed]
- Jacobs LA, Pucci DA. Adult survivors of childhood cancer: the medical and psychosocial late effects of cancer treatment and the impact on sexual and reproductive health. J Sex Med 2013;10:120-6. [PubMed]
- Bober SL, Zhou ES, Chen B, et al. Sexual function in childhood cancer survivors: a report from Project REACH. J Sex Med 2013;10:2084-93. [PubMed]
- Kenney LB, Cohen LE, Shnorhavorian M, et al. Male reproductive health after childhood, adolescent, and young adult cancers: a report from the Children’s Oncology Group. J Clin Oncol 2012;30:3408-16. [PubMed]
- Zebrack BJ, Casillas J, Nohr L, et al. Fertility issues for young adult survivors of childhood cancer. Psychooncology 2004;13:689-99. [PubMed]
- Noll RB, Bukowski WM, Rogosch FA, et al. Social interactions between children with cancer and their peers: teacher ratings. J Pediatr Psychol 1990;15:43-56. [PubMed]
- Gurney JG, Krull KR, Kadan-Lottick N, et al. Social outcomes in the Childhood Cancer Survivor Study cohort. J Clin Oncol 2009;27:2390-5. [PubMed]
- Ropponen P, Siimes MA, Rautonen J, et al. Psychosexual problems in male childhood malignancy survivors. Acta Psychiatr Scand 1992;85:143-6. [PubMed]
- Puukko LR, Hirvonen E, Aalberg V, et al. Sexuality of young women surviving leukaemia. Arch Dis Child 1997;76:197-202. [PubMed]
- van Dijk EM, van Dulmen-den Broeder E, Kaspers GJ, et al. Psychosexual functioning of childhood cancer survivors. Psychooncology 2008;17:506-11. [PubMed]
- Sundberg KK, Lampic C, Arvidson J, et al. Sexual function and experience among long-term survivors of childhood cancer. Eur J Cancer 2011;47:397-403. [PubMed]
- Hall AE, Boyes AW, Bowman J, et al. Young adult cancer survivors’ psychosocial well-being: a cross-sectional study assessing quality of life, unmet needs, and health behaviors. Support Care Cancer 2012;20:1333-41. [PubMed]
- Park ER, Norris RL, Bober SL. Sexual health communication during cancer care: barriers and recommendations. Cancer J 2009;15:74-7. [PubMed]
- Cox A, Jenkins V, Catt S, et al. Information needs and experiences: an audit of UK cancer patients. Eur J Oncol Nurs 2006;10:263-72. [PubMed]
- Stead ML, Fallowfield L, Brown JM, et al. Communication about sexual problems and sexual concerns in ovarian cancer: qualitative study. BMJ 2001;323:836-7. [PubMed]
- Bruner DW, Boyd CP. Assessing women’s sexuality after cancer therapy: checking assumptions with the focus group technique. Cancer Nurs 1999;22:438-47. [PubMed]
- Barni S, Mondin R. Sexual dysfunction in treated breast cancer patients. Ann Oncol 1997;8:149-53. [PubMed]
- Catt S, Fallowfield L, Jenkins V, et al. The informational roles and psychological health of members of 10 oncology multidisciplinary teams in the UK. Br J Cancer 2005;93:1092-7. [PubMed]
- Wiggins DL, Wood R, Granai CO, et al. Sex, intimacy, and the gynecologic oncologists: survey results of the New England Association of Gynecologic Oncologists (NEAGO). J Psychosoc Oncol 2007;25:61-70. [PubMed]
- Stead ML, Brown JM, Fallowfield L, et al. Lack of communication between healthcare professionals and women with ovarian cancer about sexual issues. Br J Cancer 2003;88:666-71. [PubMed]
- Bober SL, Recklitis CJ, Campbell EG, et al. Caring for cancer survivors: a survey of primary care physicians. Cancer 2009;115:4409-18. [PubMed]
- Hordern AJ, Street AF. Communicating about patient sexuality and intimacy after cancer: mismatched expectations and unmet needs. Med J Aust 2007;186:224-7. [PubMed]
- Hordern AJ, Street AF. Constructions of sexuality and intimacy after cancer: patient and health professional perspectives. Soc Sci Med 2007;64:1704-18. [PubMed]
- Schover LR, Brey K, Lichtin A, et al. Oncologists’ attitudes and practices regarding banking sperm before cancer treatment. J Clin Oncol 2002;20:1890-7. [PubMed]
- Juraskova I, Butow P, Robertson R, et al. Post-treatment sexual adjustment following cervical and endometrial cancer: a qualitative insight. Psychooncology 2003;12:267-79. [PubMed]
- Hartmann U, Burkart M. Erectile dysfunctions in patient-physician communication: optimized strategies for addressing sexual issues and the benefit of using a patient questionnaire. J Sex Med 2007;4:38-46. [PubMed]
- Butler L, Banfield V, Sveinson T, et al. Conceptualizing sexual health in cancer care. West J Nurs Res 1998;20:683-99. [PubMed]
- Hack TF, Degner LF, Parker PA. The communication goals and needs of cancer patients: a review. Psychooncology 2005;14:831-45. [PubMed]
- Mallinger JB, Griggs JJ, Shields CG. Patient-centered care and breast cancer survivors’ satisfaction with information. Patient Educ Couns 2005;57:342-9. [PubMed]
- Schover L, Jensen SB. eds. Sexuality and chronic illness: a comprehensive approach. New York, NY: The Guilford Press, 1988.
- Schover LR. eds. Sexuality and fertility after cancer. New York: John Wiley & Sons, 1997.
- Tsimtsiou Z, Hatzimouratidis K, Nakopoulou E, et al. Predictors of physicians’ involvement in addressing sexual health issues. J Sex Med 2006;3:583-8. [PubMed]
- Bober SL, Carter J, Falk S. Addressing female sexual function after cancer by internists and primary care providers. J Sex Med 2013;10:112-9. [PubMed]
- Rosen R, Brown C, Heiman J, et al. The Female Sexual Function Index (FSFI): a multidimensional self-report instrument for the assessment of female sexual function. J Sex Marital Ther 2000;26:191-208. [PubMed]
- Corona G, Jannini EA, Maggi M. Inventories for male and female sexual dysfunctions. Int J Impot Res 2006;18:236-50. [PubMed]
- Jeffery DD, Tzeng JP, Keefe FJ, et al. Initial report of the cancer Patient-Reported Outcomes Measurement Information System (PROMIS) sexual function committee: review of sexual function measures and domains used in oncology. Cancer 2009;115:1142-53. [PubMed]
- Baser RE, Li Y, Carter J. Psychometric validation of the Female Sexual Function Index (FSFI) in cancer survivors. Cancer 2012;118:4606-18. [PubMed]
- Isidori AM, Pozza C, Esposito K, et al. Development and validation of a 6-item version of the female sexual function index (FSFI) as a diagnostic tool for female sexual dysfunction. J Sex Med 2010;7:1139-46. [PubMed]
- Rosen RC, Riley A, Wagner G, et al. The international index of erectile function (IIEF): a multidimensional scale for assessment of erectile dysfunction. Urology 1997;49:822-30. [PubMed]
- Rosen RC, Cappelleri JC, Smith MD, et al. Development and evaluation of an abridged, 5-item version of the International Index of Erectile Function (IIEF-5) as a diagnostic tool for erectile dysfunction. Int J Impot Res 1999;11:319-26. [PubMed]
- Hatzichristou D, Rosen RC, Derogatis LR, et al. Recommendations for the clinical evaluation of men and women with sexual dysfunction. J Sex Med 2010;7:337-48. [PubMed]
- Sadovsky R, Mulhall JP. The potential value of erectile dysfunction inquiry and management. Int J Clin Pract 2003;57:601-8. [PubMed]
- Carter J, Goldfrank D, Schover LR. Simple strategies for vaginal health promotion in cancer survivors. J Sex Med 2011;8:549-59. [PubMed]