Initial experience with Allium™ stent in the management of bulbar urethral stricture
Introduction
Stents have revolutionised the management of occlusive urological diseases. Ureteric stents are commonly used in the management of various upper tract conditions. There is ongoing research into whether stenting could similarly be applied in the management of urethral stenotic diseases.
Urethral strictures have an estimated incidence of 0.6–1.4% of the male population (1,2). Over 90% of urethral strictures are classified as anterior with the majority of these being located in the bulbar urethral tract (3). While the majority of bulbar urethral strictures have an undefined aetiology, (3) known causes include trauma, surgery, brachytherapy and insertion of indwelling urethral catheters. Urethral strictures cause morbidity including lower urinary tract symptoms, infections and incontinence (4).
The management of strictures depends on the length and location within the urethra. Urethroplasty is the gold standard for bulbar strictures (5) with a low recurrence rate. Breyer et al. found 21% of patients had stricture recurrence after urethroplasty (6). However, minimally invasive alternatives may be preferable for certain patient populations. The most utilised minimally invasive interventions are urethral dilatation and internal urethrotomy (2) which have comparable outcomes, with greatest efficacy in uncomplicated strictures less than 2 cm (7). These techniques have disappointingly high recurrence rates with studies finding 58% and 92% recurrence at long-term follow up (8,9).
In 1980 urethral stents were proposed to optimise the management of urethral strictures (10). The ideal urethral stent would be easily inserted and removed, allowing urine to bypass any urethral stenosis, while maintaining the integrity of the urethral sphincters. Once in place it should not migrate, calcify, cause pain or become infected. Various iterations have been developed to ameliorate stent failure and reduce complication rates. Stent design varied in shape, proportion, material composition and incorporated special features including thermodynamic structure, gold-plating and anchors. First generation stents were designed for permanent insertion, aiming to epithelialized within the urethra. Poor long term results and difficulty reversing complications resulted in them having limited uptake and after many years stents fell out of use (11). Consequently, temporary stents emerged as the logical design for urethral stents. The study assesses the performance of the Allium™ Bulbar Urethral Stent, a second generation temporary stent, by a single surgeon across multiple Australian hospitals.
Methods
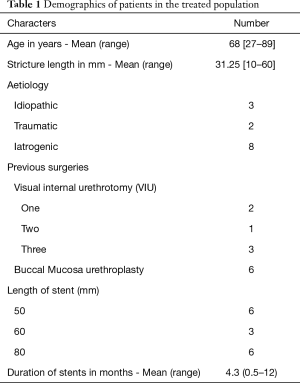
The Allium™ Bulbar Urethral Stent (BUS) is a temporary single-use stent designed for treating bulbar urethral strictures (Figure 1). It is a second generation stent composed of a nitinol metal coil with a polymer coating. The coating is designed to prevent epithelialisation and mucosal hyperplasia post urethrotomy, thereby maintaining patency and reducing stricture recurrence (12). The stents are produced in three lengths, 50, 60, and 80 mm, all of which are 45 French in calibre. There is a dynamic segment which tapers in force and radial diameter to maintain function of the external urethral sphincter. An optical element of a cystoscope is utilised for the deployment of the stent: using direct visualisation, the stent is advanced up to the external sphincter. Once in position, a trigger on the deployment device is pulled, releasing the stent. Thereafter, the stent self-expands into a cylindrical shape, matching the anatomy of the normal bulbar urethra and exerting an outward force on the urethral wall. All patients had optical urethrotomy prior to insertion of BUS. Four patients had suprapubic catheter (SPC) insertion prior to BUS placement.

A retrospective analysis was undertaken of the outcomes from a single surgeon’s experience with Allium™ BUS. Fifteen stents were placed in a total of 13 patients between 2014 and 2016. The stents ranging from 50–80 mm were inserted for between 2 weeks and 18 months. The data collated combined patient demographics, stricture length and aetiology, previous surgeries and duration of stent insertion (Table 1). The success criteria after removal of stent were voiding flow test 15 mL/sec or greater with no evidence of stricture on endoscopy or urethrogram.
Full table
Results
Allium™ stents were inserted successfully in all patients at the desired position. All patients except five were discharged on the same day after passing urine satisfactorily. Four patients had suprapubic catheter insertion prior to BUS placement and required an overnight stay. SPC was removed next day in three patients after successful voiding. One patient with SPC could not void and required permanent SPC due to his frail medical condition. One patient had haematuria post-operatively and difficulty in voiding. He was managed by gentle insertion of 12F Foley periurethral catheter for 24 hours.
We intended to keep stents indwelling for 12 months, as recommended by Allium™, but the duration of stenting varied significantly depending upon the clinical response, with a mean duration of 4.3 months. Eight of the stents were considered successful management for the patients’ bulbar urethral stricture disease as per the defined success criteria of voiding flow test 15 mL/sec or more with no evidence of stricture on endoscopy or urethrogram with a mean follow up 7.2 months (range, 2–12 month).
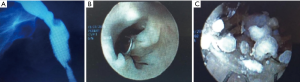
Seven stents were considered failures, however two of these failures were subsequently corrected with an exchange of urethral stents. In one case the failure was due to a distal stricture and was corrected by replacement with a longer stent. In the other case requiring reinsertion, the initial stent was passed through voiding on day 1 post insertion. Reinsertion of the same length stent yielded a successful outcome. Of the five other failed stents, two stents were buried distally into mucosa. One stent upon removal was blocked with encrustations (Figure 2). One stent migrated distally and failed. One stent had a stricture beyond the stent end.
Of the failure group, three patients were unable to void: one required a permanent SPC, another continued to intermittently self dilate, while another presented with acute urinary retention two weeks postoperatively due to stent migration. Two patients were also considered to have failed procedures due to extremely low flow rates (Qmax 7.6 and 11 mL/sec), both received further VIU.
Discussion
Despite the evolution of stent designs, complications continue to occur and therefore limit stent use. The first generation permanent stents had issues with encrustation, migration and stone formation (13). The technical difficulty of removing permanent stents after failure renders them a non-viable option.
Temporary stents may achieve lumen patency for periods greater than dilatation and repeat urethrotomy. However, temporary stents may also migrate, encrust, become infected or cause irritative symptoms including dysuria and haematuria (14).
The Allium™ BUS was eventually successful in over 60% of patients assessed in this cohort, who had previously failed other therapy. This is compared to results from Culha et al. which noted a success rate of 75% as measured by no evidence of stricture and a “significant increase in peak urinary flow”. In their multi-centre study of 64 patients, with a mean age 25 years younger than our cohort, significant improvements were seen in patients with strictures less than 2 cm and a stent duration of 9 months or greater (12). In our treated population there were high rates of stent encrustation and distal burying, occurring 20% of stent insertions. Compared with Culha et al.’s trial, a higher percentage of patients in our study achieved a peak urinary flow ≥15 mL/sec. Overall, these findings are not as positive as results in other countries as stated above (15).
The patients involved in this cohort often brought a greater disease burden, either being unable to undertake first line treatment due to being medically unfit or having already failed previous interventions. In a difficult cohort by virtue of significant comorbidity, some patients did have demonstrable improvement with the BUS, even when urethroplasty was either unsuccessful or unsuitable. Herein lies the limited role this therapy may offer in some patients who have either failed or are ineligible for first line options. However, the significant failure rate of bulbar urethral stenting limits its utility and until this is overcome, it will remain a second or third line interventional management option.
Conclusions
Urethral stents are a slowly emerging form of managing urethral strictures in patients that cannot tolerate or fail a urethroplasty. Generations of urethral stents have become more sophisticated, leading to reduced complication and failure rates. Presently temporary stenting appears to be the more efficacious approach to stent insertion. Beyond urethroplasty, the Allium™ Bulbar Urethral Stent may provide an alternative for patients to self-catheterisation, sounding or VIU.
Our experience with the Allium™ stent has been mixed, however the patient cohort assessed had significant stenotic disease, mostly having already failed urethroplasty and three patients had brachytherapy induced stricture disease. Nonetheless, some patients have received successful procedures with satisfactory flow post removal of their stents.
While there is mixed evidence for this new form of urethral stenting, we would argue it has a place for urologists in certain patients. It is difficult to make any definitive recommendation for the use of BUS due to our short duration of follow-up in a small series of disparate patients. It may have some role in patients, who are unfit for definitive procedure, radiation strictures that are not suitable for urethroplasty and in select patients who have failed urethroplasty. Further research, including longer term follow up with repeat stenting, and continued development is required to refine the Allium™ BUS in order to outperform other minimally invasive options of managing bulbar urethral strictures.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Santucci RA, Joyce GF, Wise M. Male urethral stricture disease. J Urol 2007;177:1667-74. [Crossref] [PubMed]
- Anger JT, Buckley JC, Santucci RA, et al. Trends in stricture management among male Medicare beneficiaries: underuse of urethroplasty? Urology 2011;77:481-5. [Crossref] [PubMed]
- Palminteri E, Berdondini E, Verze P, et al. Contemporary urethral stricture characteristics in the developed world. Urology 2013;81:191-6. [Crossref] [PubMed]
- Anger JT, Santucci R, Grossberg AL, et al. The morbidity of urethral stricture disease among male medicare beneficiaries. BMC Urol 2010;10:3. [Crossref] [PubMed]
- Greenwell TJ, Venn SN, Mundy AR. Changing practice in anterior urethroplasty. BJU Int 1999;83:631-5. [Crossref] [PubMed]
- Breyer BN, McAninch JW, Whitson JM, et al. Multivariate analysis of risk factors for long-term urethroplasty outcome. J Urol 2010;183:613-7. [Crossref] [PubMed]
- Steenkamp JW, Heyns CF, de Kock ML. Internal urethrotomy versus dilation as treatment for male urethral strictures: a prospective, randomized comparison. J Urol 1997;157:98-101. [Crossref] [PubMed]
- Pansadoro V, Emiliozzi P. Internal urethrotomy in the management of anterior urethral strictures: long-term followup. J Urol 1996;156:73-5. [Crossref] [PubMed]
- Al Taweel W, Seyam R. Visual Internal Urethrotomy for Adult Male Urethral Stricture Has Poor Long-Term Results. Adv Urol 2015;2015:656459. [Crossref] [PubMed]
- Fabian KM. The intra-prostatic "partial catheter" (urological spiral) (author's transl). Urologe A 1980;19:236-8. [PubMed]
- Bahouth Z, Meyer G, Yildiz G, et al. Update in urethral stents. Arch Esp Urol 2016;69:601-6. [PubMed]
- Culha M, Bahouth Z, Ozkuvanci U, et al. AlliumTM Bulbar Urethral Stent: An Updated Long-Term Multi-Center Study with New Treatment Modality for Bulbar Urethral Stricture. Open Journal of Urology 2016;6:43-8. [Crossref]
- Hussain M, Greenwell TJ, Shah J, et al. Long-term results of a self-expanding wallstent in the treatment of urethral stricture. BJU Int 2004;94:1037-9. [Crossref] [PubMed]
- Temeltas G, Ucer O, Yuksel MB, et al. The long-term results of temporary urethral stent placement for the treatment of recurrent bulbar urethral stricture disease? Int Braz J Urol 2016;42:351-5. [Crossref] [PubMed]
- Culha M, Ozkuvanci U, Ciftci S, et al. Management of recurrent bulbar urethral stricture-a 54 patients study with Allium bulbar urethral stent (BUS). Int J Clin Exp Med 2014;7:3415-9. [PubMed]


