Phosphodiesterase inhibitors for the management of lower urinary tract symptoms in men without benign prostatic hyperplasia: a systematic review
Highlight box
Key findings
• Five clinical trials assessed the efficacy of phosphodiesterase type 5 inhibitors (PDE5-Is) for lower urinary tract symptoms (LUTS) unrelated to benign prostatic hyperplasia (BPH).
• Tadalafil significantly improved urinary symptoms (both storage and voiding), pain, sexual function, and overall quality of life and patient satisfaction across multiple LUTS etiologies (post-brachytherapy, double-J stent-related symptoms, and overactive bladder).
• Tadalafil 5–10 mg daily showed similar or superior efficacy compared to alpha blockers in most studies.
• Combining tadalafil with tamsulosin or solifenacin showed enhanced benefits for patients.
• PDE5-Is are well-tolerated with minimal side effects.
What is known and what is new?
• It is known that phosphodiesterase type 5 inhibitors are beneficial in the treatment of BPH-related sexual dysfunction and urinary symptoms.
• This review highlights the benefits in treating non-BPH LUTS in men and highlights symptomatic and quality of life benefits beyond improvements in urinary parameters.
What is the implication, and what should change now?
• Tadalafil can be considered as a first-line or adjunct therapy to treat LUTS that are unrelated to BPH in men.
• Large, well-designed randomized controlled trials are needed to confirm long-term efficacy, safety, and highlight the physiological mechanisms of tadalafil in treating LUTS.
Introduction
Male lower urinary tract symptoms (LUTS) are common, and they can significantly affect patients’ quality of life. It is estimated that up to 41% of male individuals over the age of 40 years have bothersome symptoms. In men, LUTS can result from a variety of conditions, with benign prostatic obstruction (BPO) and/or overactive bladder (OAB) being two of the most prevalent causes of LUTS in male patients (1). LUTS is defined by The European Association of Urology (EAU) and American Urological Association (AUA) guidelines as either storage (irritative) symptoms (including: daytime urinary frequency, urgency, and nocturia), voiding (obstructive) symptoms (straining, weak stream, intermittency, and incomplete emptying), or postmicturition symptoms (postmicturition dribbling) affecting the lower urinary tract (LUT) (2). Bothersome LUTS are quite common after prostate brachytherapy which can substantially impair quality of life (3). Although stent discomfort can vary across patients, it is believed to affect over 80% of patients with ureteral stents. The symptoms related to ureteral stents include storage symptoms such as urinary frequency, urgency, dysuria, incomplete emptying, flank and suprapubic pain, urinary incontinence, and hematuria (4).
Phosphodiesterase type 5 inhibitors (PDE5-Is) are well-known medications in the management of erectile dysfunction (ED). They work by causing muscle relaxation and penile vasodilation by increasing cGMP levels, leading to efficient erection. Their therapeutic potential extends beyond ED, with clinical approval for treating benign prostatic hyperplasia (BPH) and LUTS (5).
This study aims to evaluate the efficacy and safety of PDE5-Is in men with LUTS unrelated to BPH to clarify their therapeutic potential in understudied populations and address current knowledge gaps. We present this article in accordance with the PRISMA reporting checklist (available at https://tau.amegroups.com/article/view/10.21037/tau-2025-aw-788/rc) (6).
Methods
The protocol for this systematic review was registered on PROSPERO in advance, where the protocol can be accessed (CRD420251072896) (7).
Study strategy and study selection
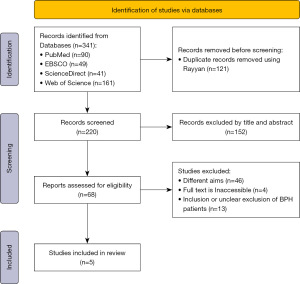
We searched four different electronic databases, which are: PubMed, EBSCO, Web of Science, and ScienceDirect, for articles published from inception until June 2025. Search terms consisted of the following terms: (“Phosphodiesterase 5 Inhibitor*” OR “PDE5 inhibitor*” OR Sildenafil OR Tadalafil OR Vardenafil OR Avanafil) AND (“Lower Urinary Tract Symptom*” OR LUTS) NOT (“Benign Prostatic Hyperplasia” OR BPH OR “Prostate Enlargement” OR “Prostatic Hyperplasia”). Search was mainly done in title and abstract. Articles were uploaded and managed by Rayyan AI software for screening (8). Study selection and screening was achieved using a three-step approach: title, abstract, and full text by two authors, and conflict was resolved by a third one. Selection of studies was done according to the following eligibility criteria: (I) published in English; (II) articles that are accessible in full text, or if provided by the authors as we contacted them in missing relevant articles; (III) only studies on humans; (IV) clinical trials; (V) studies published from inception to June 1st, 2025; (VI) studies with clear exclusion criteria for BPH, whether they excluded BPH by prostate volume cutoff value or prostate specific antigen (PSA) levels; (VII) studies done on male individuals; (VIII) studies involving at least one group receiving a PDE5 agent as a monotherapy or combined with another agent. All articles that were in foreign languages, unavailable in full text, observational designs or reviews, or those which did not specify the exclusion of BPH patients or included mixed populations without separate reporting of data according to gender were excluded from this review. Several randomized trials were excluded because they included BPH patients or mixed-sex populations. A PRISMA flowchart demonstrating the selection process is shown in Figure 1.
Data extraction
The study characteristics and outcomes data were extracted and recorded using Microsoft Excel. Data extraction was done independently by two authors. Discrepancies in data extraction were resolved by reference to the original articles and through discussion. Missing information was obtained by contacting the corresponding author of the study. Primary outcomes were subjective symptoms (pain/discomfort, voiding and storage symptoms, and sexual function), objectives measures [post-void residual (PVR) volume, urinary flow rate, voided volume], and symptom scores such as: Overactive Bladder Symptom Score (OABSS), International Index of Erectile Function (IIEF-5), and IPSS. Secondary outcomes included reported side effects and patient satisfaction. Extracted data from each study included the following items: last name of the first author, year of publication, location of the study, objectives, sample size, study groups, indication for PDE5-Is, intervention given (agents, doses, frequency, and duration), follow up period, outcome-related details (symptoms studied, scores used to define severity of symptoms, and patient-related satisfaction), author-reported limitations and conclusion. Study characteristics is shown in Table 1.
Table 1
| Study | Location | Study design | Sample size | Mean age (years) | Indication of PDE5 inhibitors | Intervention given and follow up | Outcomes measured |
|---|---|---|---|---|---|---|---|
| Yu et al. (2016) (9) | Korea | Non-randomized trial | 212 patients were initially involved; 80 patients were selected for the prospective clinical analysis | Group A: 65.3±7.9, Group B: 67.4±5.4 | LUTS after brachytherapy in prostate cancer patients | All 80 patients received tamsulosin 0.4 mg as monotherapy until 1 month after the implantation. Patients were then divided into two groups: 45 patients received tamsulosin 0.4 mg monotherapy, and the other 35 received tamsulosin 0.4 mg plus sildenafil 25 mg combination therapy until 3 months post-implantation due to the presence of ED | LUTS were compared between the two groups using the IPSS, Qmax and the pre-implantation PVR volume at 1 and 3 months after implantation. IIEF-5 was also measured for the combination group |
| Matsuo et al. (2020) (10) | Japan | Open-label, non-randomized trial | 53 male patients | 72.1±7.2 | LUTS due to OAB | The patients received 5 mg tadalafil orally once daily, and their symptoms were assessed before and after the 12-week treatment | OABSS and IPSS were used to evaluate the subjective symptoms. Uroflowmetry was used to evaluate the objective findings. Oxidative stress was assessed by determining urinary levels of 8-hydroxy-2'-deoxyguanosine levels with an adjustment for urinary creatinine concentration |
| Minagawa et al. (2021) (11) | Japan | Open-label, randomized trial | 107 patients enrolled into the study; 96 patients were analyzed | 66.1±6.8 | LUTS after brachytherapy in prostate cancer patients | Eligible patients were randomized in a 1:1 ratio to a tadalafil group (5 mg/day) or a tamsulosin group (0.2 mg/day). Tadalafil and tamsulosin were administered from the day after low-dose-rate implantation for a period of 1 year | Primary outcome: IPSS score; secondary outcomes: quality of life index, OABSS, Qmax, PVR, Sexual Health Inventory for Men score, and PSA level |
| Palinrungi et al. (2023) (12) | Indonesia | Double-blinded randomized controlled trial | 161 patients | Not specified | LUTS due to DJ ureteral stent | Mirabegron 50 mg (Group A), solifenacin 5 mg (Group B), tamsulosin 0.4 mg (Group C), tadalafil 10 mg (Group D), mirabegron 25 mg + solifenacin 5 mg (Group E), tamsulosin 0.4 mg + solifenacin 5 mg (group F), and placebo (Group G). Follow up for 4 weeks | The Ureteral Stent Symptoms Questionnaire was used to compare treatment outcomes |
| Ilyas et al. (2021) (13) | Indonesia | Double-blinded randomized controlled trials | 50 patients (25 per group) | Not specified | LUTS due to DJ ureteral stent | Group 1: tadalafil 10 mg daily, group 2: tamsulosin 0.4 mg daily. Follow up for 4 weeks. | Voiding symptoms score, pain score, general condition score, work activity score, sexual activity score, and additional symptoms score |
DJ, double-J; ED, erectile dysfunction; IIEF-5, International Index of Erectile Function-5; IPSS, International Prostate Symptom Score; LUTS, lower urinary tract symptoms; OAB, overactive bladder; OABSS, Overactive Bladder Symptom Score; PSA, prostate-specific antigen; PVR, post-void residual; Qmax, maximum urinary flow rate.
Risk of bias assessment
The process of risk of bias assessment was done using Cochrane Risk of Bias 2 (RoB-2) tool for randomized trials, and ROBINS-I for non-randomized trials (14,15). It was conducted independently by three authors and discrepancies were resolved through discussion (Figures 2,3).


Data synthesis
In this study, quantitative description and combination of outcomes were intended initially to compare the efficacy of phosphodiesterase inhibitors on LUTS. However, given the limited number of eligible studies and the heterogeneity among the studies’ groups and interventions, a quantitative meta-analysis was not feasible. Thus, an analytical descriptive synthesis of data was utilized for the purposes of this review.
Results
Overview of the included studies
A comprehensive search across four electronic databases (PubMed, EBSCO, ScienceDirect, and Web of Science). Only five studies met all the inclusion criteria and were incorporated into the final systematic review. The entire selection process is systematically outlined in the PRISMA flow diagram (Figure 1).
Included studies were published between 2016 and 2023 and emanated from three different countries, including Korea, Japan, and Indonesia. With regard to the study design, all studies were clinical trials.
LUTS due to double-J (DJ) stent insertion
Two different trials evaluated the efficacy of LUTS due to DJ stent insertion. In the study of Ilyas et al. (13), 50 patients who had symptoms related to a ureteral stent were split into two groups. Group I had 25 patients who took 10 mg of tadalafil every day, and Group II had 25 patients who took 0.4 mg of tamsulosin every day. The main measure used to assess the results was the Ureteral Symptoms Score Questionnaire (USSQ). This questionnaire looked at six areas: voiding symptoms, pain, general condition, work activity, sexual activity, and additional symptoms. The results showed that tadalafil demonstrated superior efficacy compared to tamsulosin at reducing most of the symptoms related to the ureteral stent. Specifically, tadalafil patients showed significant reduction in voiding symptoms scores by weeks 3 and 4 compared to those taking tamsulosin (P<0.001). Pain levels also significantly improved for tadalafil patients throughout all the weeks (P<0.001). For general condition, tadalafil improved symptoms more in weeks 3 and 4 (P<0.001), but earlier weeks didn’t show a big difference. Work activity and sexual activity scores improved significantly for tadalafil patients in all weeks (P<0.001). Additionally, tadalafil started to reduce additional symptoms more effectively from week 2 onwards (P<0.001). These results suggest that while both medicines were helpful, tadalafil was better at reducing symptoms and improving the quality of life for patients with ureteral stent-related symptoms.
In another prospective, randomized clinical trial by Palinrungi et al. (12), 161 patients with LUTS associated with DJ ureteral stents were enrolled. Participants were randomly distributed into seven intervention groups: mirabegron 50 mg (Group A), solifenacin 5 mg (Group B), tamsulosin 0.4 mg (Group C), tadalafil 10 mg (Group D), a combination of mirabegron 25 mg and solifenacin 5 mg (Group E), tamsulosin 0.4 mg combined with solifenacin 5 mg (Group F), and placebo (Group G). Eligible patients were aged between 18 and 79 years, had ureteral stones measuring less than 10 mm, and had undergone endoscopic stent insertion. Individuals with comorbid conditions such as malignancy, urinary tract infection, diabetes mellitus, cardiovascular disorders, or recent surgical procedures were excluded.
Symptom outcomes were measured using the USSQ, which assesses six primary domains: urinary symptoms, pain levels, overall well-being, sexual health, work performance, and miscellaneous issues. Assessments were carried out on days 7, 14, 21, and 28 after stent insertion. Among all groups, the combination of tamsulosin and solifenacin (Group F) yielded the most notable improvement in urinary discomfort (mean score 2.5), occupational functioning (0.2), sexual health (0.0), and additional stent-related problems (0.45), indicating the broadest overall symptom relief. In contrast, tadalafil monotherapy (Group D) was most effective in reducing pain and enhancing general health status, with both domains showing the lowest scores (0.45) by the fourth week. While all treatment groups showed statistically significant reductions in LUTS compared to baseline, the combination of tamsulosin with solifenacin and tadalafil alone emerged as the most efficacious therapeutic option.
LUTS due to brachytherapy
Two trials were focused on assessing the efficacy of phosphodiesterase inhibitors on LUTS emerging from brachytherapy for the treatment of prostate cancer. Minagawa et al. (11) explored the effects of tadalafil and tamsulosin on urinary and sexual health following low-dose-rate (LDR) brachytherapy in patients with prostate cancer. A total of 96 men diagnosed with early-stage prostate cancer were enrolled and given either 5 mg of tadalafil or 0.2 mg of tamsulosin daily for one year after undergoing treatment. The main measure was the IPSS, as well as other tests like urine flow rate, PVR, and the Sexual Health Inventory for Men (SHIM) score. Both groups had a short period where their urinary symptoms got worse, usually around 3 months after treatment, but they gradually improved. Most of the time, no significant difference was observed between the two groups when it came to IPSS or the bladder function tests. However, at 9 months, tadalafil slightly improved the IPSS and the part of the score related to storing urine. Also, men on tadalafil had better sexual health, with higher SHIM scores at 6, 9, and 12 months compared to those on tamsulosin. These results show that tadalafil may be a good choice for helping with urinary issues and also for keeping sexual health strong during recovery after LDR brachytherapy.
Another study (9) evaluating the efficacy of PDE5-Is in patients with LUTS after brachytherapy found that the group receiving both tamsulosin and sildenafil demonstrated a significant decrease in their ability to get and keep an erection 1 month after the procedure. Their IIEF-5 scores went down from 14.36±4.51 to 9.17±3.82, which was a big difference (P=0.003). By three months, there was some recovery, but it did not return to baseline, with scores at 11.03±3.54 (P=0.04). When it came to LUTS, both the group taking just one drug and the group taking both drugs showed improvement in their IPSS scores by the 3rd month. However, the group taking both drugs had a bigger improvement in total IPSS scores compared to the group taking just one drug (P=0.03). The combination group also had a noticeable improvement in the storage part of the IPSS (P=0.047), but the difference was not statistically significant in the voiding score (P=0.06) or the voiding-to-storage ratio (IPSS V/S, P=0.076) to be significant. Both groups showed improvement in maximum urinary flow rate (Qmax) and PVR, with the combination group showing better results, though specific values were not reported for those improvements. It should be noted that the magnitude of the treatment effects may have been influenced by the difference in the baseline group sizes of the monotherapy and combination groups.
LUTS due to OAB
One study (10) assessed the efficacy of 5 mg of daily tadalafil for 12 weeks on symptoms of OAB in 53 men. Subjective scores (OABSS and IPSS) significantly improved after tadalafil treatment, as the mean total OABSS decreased from 6.5±1.7 to 2.8±1.9 (P<0.001), with significant improvement in individual symptoms of daytime frequency, nighttime frequency, urgency, and urge incontinence (P<0.001 in all of which). Furthermore, the IPSS score was reduced from 14.4±5.7 to 7.7±3.4 (P<0.001), with a significant drop in both storage symptoms (7.3±2.8 to 3.8±2.4; P<0.001) and voiding symptoms subscores (7.1±4.8 to 4.0±2.9; P<0.001). Almost all individual IPSS domains (incomplete emptying, frequency, urgency, weak stream, straining, and nocturia) witnessed significant improvement upon tadalafil treatment, except for intermittency (P=0.07). Nonetheless, quality of life scores improved from 4.2±1.0 to 2.3±0.9 (P<0.001).
Objective findings (mean voided volume, Qmax, and PVR) also witnessed important improvements upon receiving tadalafil. The mean voided volume increased from 116.3±78.3 mL at baseline to 168.4±102.6 mL after treatment (P=0.002). Qmax also improved from 7.9±3.5 to 10.8±5.7 mL/s (P<0.001) after tadalafil treatment. Post-void residual decreased from 42.3±31.7 to 37.3±27.1 mL after treatment, but this change remained statistically insignificant (P=0.26). Overall, 12 weeks of 5 mg tadalafil treatment led to both subjective and objective improvements in male patients with OAB.
Discussion
Male LUTS are common, affecting up to 41% of men over 40 years. Common causes include BPO and/or OAB (1). LUTS can be categorized as storage, voiding, or postmicturition symptoms (2). On this basis, the present systematic review aimed to clarify the therapeutic potential of PDE5-Is in this understudied population, address existing knowledge gaps, and support more targeted, mechanism-based strategies for managing LUTS in men without prostate enlargement or obstruction.
Yu et al. (9) found that patients with LUTS after brachytherapy implantation may benefit from combination therapy with tamsulosin and low-dose sildenafil. A comparable baseline was noted before the treatment, with both groups (the monotherapy and combination therapy groups) having similar PVR volumes, maximal urine flow rates, and total IPSS. One month after implantation, both groups showed increased IPSS and PVR, likely due to postoperative changes; however, these changes were not significant between both groups. By the 3rd month, the combination therapy group showed a significant improvement in IPSS compared to the monotherapy group (P=0.03), suggesting better symptom control. Contrarily, Qmax and PVR remained similar between groups, indicating that subjective symptom improvement may not reflect changes in objective urinary measurements. In general, Yu et al. concluded that low-dose sildenafil combined with tamsulosin exhibits greater symptom relief for LUTS after brachytherapy implantation without affecting urinary flow measurements or residual volume (9). Findings of this study should be interpreted cautiously, as the effect is modest and the statistical significance may have been influenced by the baseline numerical difference between the treatment groups rather than a true clinical effect.
Matsuo et al. (10) demonstrated that tadalafil significantly improves urodynamic parameters and symptoms of OAB, likely through the reduction of oxidative stress. Administration of tadalafil led to significant improvements in the total and subcomponent scores of the OABSS, suggesting broad symptom control. Similarly, storage and voiding symptoms captured by the IPSS improved considerably. Objective measures such as Qmax and voided volume also increased significantly (P=0.002 and P<0.001, respectively), indicating better bladder emptying and urinary efficiency. These findings suggest that tadalafil improves both subjective symptoms and objective function (10). However, it should be emphasized that the evidence supporting the use of PDE5-Is in male patients with OAB is solely derived from a single non-randomized trial. Therefore, it should be considered as hypothesis-generating rather than definitive evidence.
The study conducted by Minagawa et al. (11) found that tadalafil can be a useful therapeutic option for LUTS after LDR brachytherapy, with the added benefit of maintaining sexual function. The IPSS indicated that both groups’ LUTS improved gradually over the course of the 12-month follow-up period after initially getting worse. Despite the fact that the tadalafil group’s IPSS readings were generally marginally higher at most time points, the group differences were not statistically significant, with the exception of the 9-month mark. Uroflowmetry measurements and postvoid residual urine volumes again showed no significant variations across the groups, suggesting comparable objective urinary function. The results for sexual health, however, showed a significant difference. It appears that tadalafil better maintains erectile function during the post-brachytherapy recovery phase because the tadalafil group’s SHIM values were considerably higher at 6, 9, and 12 months after treatment. Generally, tadalafil and tamsulosin both offer comparable relief for LUTS following brachytherapy; however, tadalafil has the added advantage of preserving improved sexual health, which may make it a better option for patients who are worried about their sexual function after treatment (11). It is important to note that tamsulosin was used as a comparator only, and it should not be interpreted as the universal standard of care for LUTS after brachytherapy.
According to Palinrungi et al. (12), although LUTS improved in all seven therapy groups, some treatments were noticeably more successful in particular domains. The greater efficacy of the tamsulosin and solifenacin combination among the different medications in treating difficulties with sexual function, work performance, and other relevant issues suggests a wider influence on patients’ quality of life. However, tadalafil monotherapy showed the greatest improvement in both general health and pain-related symptoms, suggesting potential benefit for individuals who are suffering from systemic discomfort or a decline in general well-being as a result of LUTS. All treatment methods resulted in symptom relief, but the combination of tamsulosin and solifenacin and tadalafil monotherapy proved to be the most successful. Depending on the patient’s main concerns, whether they were sexual, functional, or general health-related, each of these options had unique benefits (12).
Although 0.4 mg/day tamsulosin and 10 mg/day tadalafil both effectively reduced LUTS, Ilyas et al. (13) found that tadalafil produced better overall results in most symptom areas. Various components of symptoms were assessed, including sexual activity, work ability, pain, general condition, voiding symptoms, and additional symptoms. Tadalafil has shown more significant decreases in pain, symptoms associated with sexual activity, and other symptoms, indicating a more comprehensive effect on patients’ quality of life. Although tamsulosin had less evident effects on pain and sexual activity, it did show positive advantages, especially in reducing voiding symptoms and enhancing job competence. According to the study findings, tadalafil outperformed tamsulosin in the majority of measurable outcomes; nevertheless, there were no appreciable differences between the two therapies in terms of general condition in weeks two and four or voiding symptoms in week two. The study noted that although both drugs relieved LUTS, daily 10 mg doses of tadalafil provided more thorough symptom alleviation than tamsulosin, making it a better overall therapy option for patients with a variety of LUTS-related complaints (13).
One meta-analysis by Kallidonis et al. (16) tested the efficacy of combined alpha blockers with phosphodiesterase inhibitors for the management of ED and LUTS, and it found that the combination treatment is more effective than monotherapies for improving the IPSS scores, especially voiding subscores. However, less significant improvement is found on the Qmax and PVR (16). Furthermore, another systematic review investigating the efficacy of phosphodiesterase inhibitors on LUTS secondary to BPH concluded similar results, with tadalafil 5 mg per day being able to significantly improve the IPSS and IIEF scores, but not the mean Qmax. Also, it has been found to have patient-reported improvement in nocturnal voiding frequency and LUTS (17).
According to a meta-analysis by Sun et al. (18), it was revealed that tadalafil reduces the risk of abnormal ejaculation in lower ureter stones more than α1-adrenoceptor blockers do. Stone expulsion rates are higher and expulsion times are shorter with combination treatment than with tamsulosin alone. For LUTS, α1-adrenoceptor blockers are more effective in reducing the PVR volume and the IPSS. Combination therapy offers the most comprehensive treatment effect for both conditions (18). A meta-analysis by Wang et al. (19), comparing seven treatment approaches for LUTS associated with BPH (LUTS/BPH), found that PDE5-Is and α-blockers consistently decreased overall IPSS scores as well as storage and voiding subscores. Alpha-blockers + 5α-reductase inhibitors were the most effective combination for reducing storage symptoms. The study also revealed that the response of Qmax to PDE5-Is alone was better than that of LUTS/BPH (19).
One of our review’s main strengths is that it examines a different pharmaceutical option for male LUTS that is unrelated to prostatic enlargement. Our focus is to evaluate the efficacy of PDE5-Is—namely tadalafil—on a variety of symptoms, such as discomfort/pain, storage and voiding symptoms, and sexual dysfunction. Our goal is to address the knowledge gap by targeting understudied populations (e.g., post-brachytherapy patients) and including both subjective and objective outcomes in order to improve the clinical significance of this paper. However, the ability to meta-analyze the data and generalize the findings is limited by the small sample sizes, different follow-up periods, and heterogeneity in study designs. Furthermore, generalizing the findings is further limited by the scarcity of well-established safety data and the small number of randomized controlled trials. Despite this, tadalafil appears to have a more significant therapeutic benefit compared to traditional alpha-blockers, especially for men with LUTS unrelated to prostatic enlargement. This encourages a more individualized, mechanism-based approach to therapy. However, to validate and generalize these findings, larger and well-structured trials are required.
Conclusions
According to the findings of this systematic review, tadalafil, either alone or in combination, is an effective and well-tolerated treatment for LUTS in men who do not have prostate enlargement or obstruction. It provides advantages beyond the alleviation of urinary symptoms, such as enhancements in sexual function, pain, and overall health. To improve overall symptom control and quality of life, this study advises clinicians to consider tadalafil as a first-line or adjunct treatment for LUTS in males, especially when ED or non-obstructive reasons are also present. Furthermore, it recommends that in order to assess the long-term effectiveness, safety, and underlying mechanisms of PDE5-Is in a variety of LUTS populations, future research should concentrate on large-scale, long-term randomized controlled trials with standardized outcome measures.
Acknowledgments
None.
Footnote
Reporting Checklist: The authors have completed the PRISMA reporting checklist. Available at https://tau.amegroups.com/article/view/10.21037/tau-2025-aw-788/rc
Peer Review File: Available at https://tau.amegroups.com/article/view/10.21037/tau-2025-aw-788/prf
Funding: None.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tau.amegroups.com/article/view/10.21037/tau-2025-aw-788/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Abdelmoteleb H, Jefferies ER, Drake MJ. Assessment and management of male lower urinary tract symptoms (LUTS). Int J Surg 2016;25:164-71. [Crossref] [PubMed]
- Park HJ, Won JE, Sorsaburu S, et al. Urinary Tract Symptoms (LUTS) Secondary to Benign Prostatic Hyperplasia (BPH) and LUTS/BPH with Erectile Dysfunction in Asian Men: A Systematic Review Focusing on Tadalafil. World J Mens Health 2013;31:193-207. [Crossref] [PubMed]
- Jacobs BL, Smith RP, Beriwal S, et al. Changes in lower urinary tract symptoms after prostate brachytherapy. J Contemp Brachytherapy 2011;3:115-20. [Crossref] [PubMed]
- Miyaoka R, Monga M. Ureteral stent discomfort: Etiology and management. Indian J Urol 2009;25:455-60. [Crossref] [PubMed]
- ElHady AK, El-Gamil DS, Abdel-Halim M, et al. Advancements in Phosphodiesterase 5 Inhibitors: Unveiling Present and Future Perspectives. Pharmaceuticals (Basel) 2023;16:1266. [Crossref] [PubMed]
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 2021;372:n71. [Crossref] [PubMed]
- Booth A, Clarke M, Dooley G, et al. The nuts and bolts of PROSPERO: an international prospective register of systematic reviews. Syst Rev 2012;1:2. [Crossref] [PubMed]
- Ouzzani M, Hammady H, Fedorowicz Z, et al. Rayyan-a web and mobile app for systematic reviews. Syst Rev 2016;5:210. [Crossref] [PubMed]
- Yu YD, Kang MH, Choi CI, et al. Clinical efficacy of combination therapy with an alpha blocker and low-dose sildenafil on post-therapy lower urinary tract symptoms after low-dose-rate brachytherapy for prostate cancer. World J Urol 2016;34:1269-74. [Crossref] [PubMed]
- Matsuo T, Miyata Y, Araki K, et al. Efficacy of Tadalafil Therapy and Changes in Oxidative Stress Levels in Male Patients with Lower Urinary Tract Symptoms and Overactive Bladder. Low Urin Tract Symptoms 2020;12:47-53. [Crossref] [PubMed]
- Minagawa T, Oguchi T, Saitou T, Fukazawa A, Hashida I, Koiwai K, Iijima K, Kato H, Ogawa T, Ishizuka O. Impact of low-dose tadalafil on adverse events after low-dose-rate brachytherapy for prostate cancer: A bi-center randomized open-label trial. Int J Urol 2021;28:432-8. [Crossref] [PubMed]
- Palinrungi MA, Rasyid H, Prihantono P, et al. Double-J stent-related lower urinary tract symptoms: a prospective randomized trial of mirabegron, solifenacin, tamsulosin, and tadalafil and their combinations. Gazz Med Ital Arch Sci Med 2023;182:634-40.
- Ilyas Muh RF, Kholis K, Palinrungi MA, Palinrungi AM, et al. Comparative effectiveness of Tamsulosin and Tadalafil therapy in ureteral stents-related symptoms (uSRS). Medicina Clínica Práctica 2021;4:100226.
- Higgins JP, Altman DG, Gøtzsche PC, et al. The Cochrane Collaboration's tool for assessing risk of bias in randomised trials. BMJ 2011;343:d5928. [Crossref] [PubMed]
- Sterne JA, Hernán MA, Reeves BC, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016;355:i4919. [Crossref] [PubMed]
- Kallidonis P, Adamou C, Kotsiris D, et al. Combination Therapy with Alpha-blocker and Phosphodiesterase-5 Inhibitor for Improving Lower Urinary Tract Symptoms and Erectile Dysfunction in Comparison with Monotherapy: A Systematic Review and Meta-analysis. Eur Urol Focus 2020;6:537-58. [Crossref] [PubMed]
- Gacci M, Andersson KE, Chapple C, et al. Latest Evidence on the Use of Phosphodiesterase Type 5 Inhibitors for the Treatment of Lower Urinary Tract Symptoms Secondary to Benign Prostatic Hyperplasia. Eur Urol 2016;70:124-33. [Crossref] [PubMed]
- Sun X, Guan W, Liu H, et al. Efficacy and safety of PDE5-Is and α-1 blockers for treating lower ureteric stones or LUTS: a meta-analysis of RCTs. BMC Urol 2018;18:30. [Crossref] [PubMed]
- Wang X, Wang X, Li S, et al. Comparative effectiveness of oral drug therapies for lower urinary tract symptoms due to benign prostatic hyperplasia: a systematic review and network meta-analysis. PLoS One 2014;9:e107593. [Crossref] [PubMed]