
Characterizing the pharmacokinetics and pharmacodynamics of immunosuppressant medicines and patient outcomes in elderly renal transplant patients
Introduction
Renal transplantation is first-line treatment for patients with end stage kidney disease (ESKD), regardless of age (1,2). When compared to dialysis, renal transplantation increases life expectancy, as well as improves physical and social functioning (quality of life), mental stability, vitality, overall well-being and future perspective (2-5). The fastest growing age group with ESKD is patients aged over 65 years (4,6). In the last decade, there has been a noticeable increase in the number of elderly patients receiving a renal transplant. In the USA, the number of transplants performed in patients aged over 65 years increased from 10% to 17% of the total population between 2002 and 2012 (7). In Australia, 14% of renal transplants performed in 2015 were in patients aged 65 years of age and above (8). In Europe, a 2012 Renal Transplantation Registry report stated that transplant prevalence was 22% in the over 65 years age group and 20% in the over 75 years age group (9). This rise in the incidence of renal transplantation in the elderly can be attributed to an aging population (10), improvements in transplant outcomes (11), and the introduction of expanded criteria donor (ECD) kidneys (12).
Although age is no longer considered an absolute barrier to transplantation, long-term graft and patient survival is not improving in elderly renal transplant recipients, as compared to younger adults (1,6,10,13). The three most frequent causes of death in elderly recipients are infection, malignancy and cardiovascular events (14-16), all of which can be attributed partly to immunosuppressant medications. Studies show that older recipients have a lower risk of acute rejection due to reduced immune reactivity (15) and the elderly are the most likely age group to die with a functioning graft (1,17,18). The risk of acute rejection increases with ECD kidneys, and although elderly patients are considered at lower risk of rejection compared to younger adults, they are still at risk (19,20). Additionally, age-related comorbidities negatively influence patient outcomes in elderly recipients (13). Consequently, elderly renal transplant recipients are at greater risk of both morbidity and mortality when compared to younger recipients (21,22).
The intricate balance between under- and over-immunosuppression becomes increasingly complex in elderly recipients, due to changes in drug pharmacokinetics and pharmacodynamics (23-25). Responses seen in the elderly can be unexpected and exaggerated when compared to younger adults with similar characteristics (gender, weight, height, ethnicity) (25). Larger variability in response may be due to changes in physiology and disease, however, as studies conducted in the elderly are limited, it is difficult to draw definitive conclusions (25). Obtaining a greater understanding of immunosuppressant drug pharmacokinetics and pharmacodynamics in elderly renal transplant recipients should assist with therapeutic decisions in this unique and vulnerable sub-population. Therefore, the purpose of this review is to critique and summarize the available literature on the pharmacokinetics and pharmacodynamics of immunosuppressant medicines in elderly renal transplant recipients.
ECD kidneys
Elderly kidney transplant recipients, who receive ECD kidneys, incur additional barriers to graft survival, which compound the already significant effects of physiological aging. ECD kidneys have a shortened lifespan, with both one and five-year graft survival significantly lower than with standard donor kidneys (26). Allocation of grafts with shortened lifespans (ECD kidneys) to patients with shorter life expectancies (the elderly) is considered acceptable as the survival and quality-of-life benefits of old-for-old donor matching still outweighs long-term dialysis (14,15,17,20,27-29).
The decline in immune reactivity observed in elderly recipients is complicated by increased immunogenicity of older donor grafts (15). This has resulted in more potent early stage immune responses observed in elderly transplant recipients with ECD kidneys, which can lead to episodes of acute rejection (21) and delayed graft function (27). In one retrospective study involving 1,307 participants, it was shown that receiving an ECD kidney significantly increased the risk of acute rejection, but had no effect on overall patient survival in patients aged 60 years and over (30).
Age-related pharmacokinetic (PK) changes
A number of physiological changes associated with advancing age can affect drug absorption, distribution, metabolism and excretion in healthy adults. Decreased surface area of the intestinal epithelium, reduced splanchnic blood flow, altered gastric motility (31), delayed gastric emptying and increased gastric pH (but reduced gastric acid secretion) (31,32), can potentially lead to reduced drug absorption in the elderly. However, studies have shown that the bioavailability of drugs absorbed through permeation of the gastrointestinal tract remains largely unchanged in the elderly; with absorption of drugs via carrier-mediated transport processes most likely to be impeded (24). The absorption of basic drugs may actually improve, as the rise in gastric pH, caused by a decline in gastric parietal cells, improves absorption (24). Overall, it is assumed that there is a slight decline in drug absorption in the elderly; however, this is patient and drug specific.
Elderly patients tend to have an increased percentage of body fat, and a reduction in lean body muscle mass and total body water, compared to younger adults (24,33). These body composition changes generally occur in all elderly patients, including ‘thin’ body types (34), resulting in a volume of distribution increase for lipophilic drugs and decrease for hydrophilic drugs (31). A drug’s concentration in the plasma is inversely related to its volume of distribution, leading to a greater plasma concentration in elderly patients compared to younger adults for hydrophilic drugs. Elderly patients can also have altered drug binding to plasma proteins (albumin, alpha-1-acid glycoprotein and lipoproteins) (32,35). A reduction in serum albumin in the elderly can lead to a decrease in total drug concentration and an increase in free drug fraction, with no corresponding change in unbound (free) drug concentration. Changes in drug protein binding may be an important consideration when interpreting therapeutic drug monitoring (TDM) results for drugs where total rather than unbound drug concentration is measured (25,35).
There are a number of transporters involved in the movement of drugs across biological membranes within the body. Solute carrier (SLC) transporters mediate the uptake of drugs from the blood, transporting them across the basolateral membrane, and into cells (proximal tubule of the kidney or into hepatocytes), or the gut (into enterocytes) (36), whilst ATP-binding cassette transporters (ABC transporters) are involved in the cellular efflux of drugs and their metabolites (37). As many transporters are multi-specific, drug-drug interactions that affect drug membrane transportation can lead to altered pharmacokinetics and drug effects (37). There is some research that suggests that protein carrier activity is influenced by age, with a study by Fu, Csanaky and Curtis in 2012 showing that the mRNA levels of xenobiotic-processing genes (includes SLCs) was significantly lower in aged mice. Similarly, a study by van Assema and researchers showed that aging reduced P-glycoprotein levels in the blood-brain barrier in healthy adults (38). When considering transporter effects in the elderly, the presence and impact of multiple comorbidities or concomitant medications can affect these transporters in extremely complex ways, making it difficult to predict the final outcome.
Advancing age leads to a decline in liver size, which can cause a reduction in hepatic blood flow (approximately 40%), and reduced drug transfer across the hepatic endothelium (23,39). More specifically, drug metabolism is directly attributed to the drug’s extraction rate (high or low) by the liver, as haemoperfusion declines in the elderly; drugs with a high extraction rate have an age-related decline in clearance. Conversely, clearance of drugs with a low extraction rate remain largely unchanged, as metabolism is less dependent on hepatic blood flow than activity of membrane bound metabolising enzymes such as cytochrome P450 (CYPs) (32). Studies have shown that although it is unlikely CYP3A enzyme number or affinity reduces with age (40), drug metabolism via Phase-I CYP3A-mediated reactions may decline with advancing age (39). This is likely due to inter- morphological and circulatory changes (40), and can be variable between patients (32). Phase-II conjugation reactions (primarily glucuronidation) and the uridine-5’-diphsopho glucuronyl transferase (UGT) system (in the liver) are generally less influenced by age (32,39).
In terms of drug elimination, glomerular filtration rate (GFR) declines at a rate of approximately 1 mL/min/1.73 m2 of body surface area, from 20–30 years of age onwards (31,34). As a result, renal drug excretion is often significantly reduced in the elderly, regardless of co-morbidities. Little is known about whether biliary excretion and enterohepatic recirculation of drugs is affected by age.
A potential decline in liver, and likely decline in renal function in the elderly can cause a reduction in drug clearance, compared with younger adults. Drug clearance is the principle determinant of total drug exposure (31). Steady-state plasma drug concentration achieved for a given drug dose may be higher in elderly patients, which in turn, may increase the risk of toxicity (31). Lipophilic drugs generally have increased distribution in elderly recipients and an increased half-life. Hence drug concentrations may decline at a slower rate in the elderly (as fat acts as a reservoir for the drug) (35), increasing the likelihood of drug accumulation with multiple doses (31).
Age-related pharmacodynamic changes
Drug response is dependent on a range of factors including homeostatic mechanisms, receptor density and affinity, and signal transduction pathways (25,40,41). The entire cascade of events, from drug administration to patient response, must be considered when examining the pharmacodynamics of a drug (41). Advancing biological age leads to structural and functional changes, including decreased physiological fitness and a marked decline in the robustness of the immune system (immunosenescence) (32). The decline in functional reserve in the elderly is distinctly different between individuals (42).
With aging, there is a progressive reduction in the body’s homeostatic mechanisms. This means that in the elderly, it takes more time after administration of a pharmacological intervention (medicine) for counter-regulatory mechanisms to return the body to its original steady-state (32). Additionally, receptor number and responsiveness declines with age, however, this does not lead to a reduction in drug sensitivity or effectiveness. Instead, with advancing age, despite it taking longer for a drug to reach maximum effect (Emax), the reaction is significantly stronger, and the overall incidence of adverse effects is greater (32,33). Elderly patients may be more likely to suffer the side effects of medicines when dosed to the same target levels as younger adults.
Immunosenescence
Immunosenescence describes the reduced immune reactivity observed in elderly recipients due to: a decline in naïve T-cells (immune cells), the accumulation of memory T-cells, and alteration of B-cells, causing a decrease in antibody response (15). Immunosenescence can affect all immunological compartments and causes a shift in both the regulation and function of the total immune system (20). The primary mechanism behind this phenomenon is involution of the thymus (shrinkage of the gland responsible for the development of peripheral CD4+ and CD8+ T-cells with age), leading to a progressive reduction in the naïve T-cell count (43). Reduction in naïve T-cell number corresponds to the accumulation of memory T-cells (21,43). Inadequate output of naïve T-cells prevents homeostatic equilibrium of the peripheral T-cell pool being restored, resulting in proliferation of existing peripheral T-cells (homeostatic proliferation). However, this new population of T-cells, comprised mostly of memory T-cells, has a reduced ability to respond to new immunological challenges (21,43).
There is a paucity of evidence surrounding the impact of age on the function of B-cells (44). The literature demonstrates that advancing age affects a decline in naïve B-cells (corresponding to an increase in memory T-cells), as well as diminished B-cell antibody response (15) [compounded by the reduction in T-cells which are integral to B-cell activation (45)]. Unlike T-cells, B-cell homeostasis is maintained in the periphery through a reduction in the turnover of mature B-cells (20). Despite this, the decline in B-cells causes a change in antibody specificity, and a reduction in plasma cells in the bone marrow (20,43).
Clinically, the effects of immunosenescence in elderly renal transplant patients increases the risk of infections, malignancies, autoimmune disorders, atherosclerosis and neuro-degeneration (20,46). In transplant recipients, immunosenescence likely lowers the risk of acuterejection (47), but increases the risk of drug-related adverse effects, particularly infections and malignancy (48). However, at present, the impact of immunosenescence on the immune system response is only partly understood (49,50). Consequently, it is still unclear how best to dose immunosuppressant drugs in elderly renal transplant recipients.
Immunosuppressant medicines
The most commonly prescribed maintenance immunosuppressive protocol for kidney transplant recipients is triple therapy with a calcineurin inhibitor (cyclosporine or tacrolimus), an antimetabolite (mycophenolate or azathioprine) and a corticosteroid (prednisolone or prednisone) (51,52). This combination of drugs is prescribed concurrently, with each drug primarily targeting a different part of the immune system’s response. Tacrolimus, mycophenolate and corticosteroids is the most commonly prescribed combination, regardless of age (53,54). It is thought that intuitively, clinicians may lower immunosuppressant medicine doses in elderly patients due to their reduced immunocompetence and increased susceptibility to side effects and infection, however, the literature has yet to confirm how widespread this practice is, and how therapeutic adjustments are made (55). Currently, the dose of tacrolimus or cyclosporine is adjusted according to drug concentrations measured during TDM, whereas antimetabolites and corticosteroids are prescribed under a fixed dosing regimen in most transplant centres (51).
Calcineurin inhibitors (tacrolimus and cyclosporine)
Calcineurin inhibitors (CNI), cyclosporine and tacrolimus, block calcineurin enzymatic activity. Calcineurin phosphorylates the nuclear factors of activated T lymphocytes (T-cells) which results in the production of interleukin-2 (56). Interleukin-2 increases the proliferation and activity of T-cells, which are essential for amplification of the immune response. Blocking calcineurin prevents interleukin-2 transcription and T-cell receptor engagement (42,57), suppressing the immune system (42). It has been demonstrated in clinical trials that the rate of acute rejection at one-year post-transplant is significantly greater in patients prescribed cyclosporine compared to tacrolimus (P=0.001) (57). Hence, tacrolimus is now the more commonly prescribed CNI, and forms the foundation of 90% patients’ initial and maintenance immunosuppressive regimes (7,42).
Blockage of the calcineurin pathway affects non-immune cells, including neurons, skeletal and cardiac myocytes. Off-target effects include enhanced expression of transforming growth factor-β and development of interstitial fibrosis (a feature of nephrotoxicity), and are thought to contribute to the toxicity observed with clinical use of CNI’s (42). Both cyclosporine and tacrolimus can cause nephrotoxicity and neurotoxicity (58). However, hypertension and cosmetic complications such as hypertrichosis, gingival hyperplasia and gynaecomastia are more frequently observed in patients taking cyclosporine, whilst glucose intolerance, new-onset diabetes mellitus, alopecia, and diarrhoea impact up to 75% of patients taking tacrolimus (58).
Calcineurin inhibitors have a narrow therapeutic index and display large intra- and inter-individual pharmacokinetic variability (59-61). Over-immunosuppression (too high doses) increases the risk of side effects including neurotoxicity and nephrotoxicity, whilst under-immunosuppression increases the risk of graft rejection (59). Elderly recipients have been shown to be more susceptible to CNI side effects (27), and hence, prescribing equivalent concentrations to younger recipients may increase their risk of toxicity. Currently, specific protocols regarding the best choice and dose of CNIs in elderly recipients have not been validated (1,54).
Tacrolimus is rapidly absorbed in the gastrointestinal tract with maximum drug concentrations generally reached within 1–2 hours. It is highly lipophilic, but poorly water soluble, and passes across the intestinal wall by passive diffusion (29,45). Oral bioavailability is generally poor, with a mean value of approximately 25%, however this can vary substantially between individuals (reported range 5–95%) (42,62). Drug absorption is likely reduced by P-glycoprotein expression in the gastro-intestinal tract, as well as pre-systemic metabolism by cytochrome P450 3A (CYP3A) (62). Tacrolimus binds extensively to erythrocytes (approximately 99%), and in plasma it mainly binds to alpha-1-acid glycoprotein and albumin (62,63). For this reason, tacrolimus concentrations are typically measured in whole blood rather than plasma (42). Tacrolimus undergoes extensive metabolism via the liver and CYP3A system into more than 15 active metabolites, with >95% eliminated by the biliary route (64) and <1% unchanged drug excreted in the urine and faeces (51). Large variability in the pharmacokinetics of tacrolimus has commonly been attributed to a number of factors including cytochrome P450 genotype, drug-drug interactions, patient haematocrit, patient weight, time post-transplant and patient hepatic function (65).
Cyclosporine is highly lipophilic and only slightly water-soluble. Oral absorption is slow, and overall, cyclosporine has low oral bioavailability (median: 21%) (42), with oral absorption ranging between 10–89% (66). Drug absorption is likely reduced by P-glycoprotein expression in the gastro-intestinal tract, as well as pre-systemic metabolism by CYP3A (60). Similar to tacrolimus, cyclosporine is extensively metabolized by hepatic CYP3A into >25 metabolites and excreted predominantly by the biliary system (90%). Renal excretion accounts for only 6% of drug elimination. Cyclosporine also binds extensively to red blood cells and therefore, whole blood concentrations are typically used to measure drug concentration (42). Large variability in the pharmacokinetics of cyclosporine has been commonly attributed to a number of factors including drug-drug interactions, patient haematocrit, patient weight, time post-transplant and patient hepatic function (66).
Antimetabolites (mycophenolate)
Antimetabolites (mycophenolate and azathioprine) are critical components of maintenance immunosuppressive therapy regimens as they lower the risk of acute rejection by blocking T- and B-cell proliferation (67). Mycophenolate is the typical antimetabolite of choice, as it more effectively lowers the acute rejection risk, when compared to azathioprine (58,68). Mycophenolate is available in two forms: mycophenolate mofetil (MMF; Cellcept®) and enteric-coated mycophenolate sodium (Myfortic®) which are roughly bio-equivalent (69). Both forms are rapidly converted to the active drug, mycophenolic acid (MPA), in the body. MPA reversibly inhibits the enzyme, inosine 5'-monophosphate dehydrogenase (IMPDH). There are two isoforms of IMPDH, both of which are sensitive to inhibition by MPA (42). IMPDH plays a role in the synthesis of guanine nucleotides, which are critical to T- and B-lymphocyte proliferation and function (42). Hence, blocking the de-novo purine synthesis pathway of guanine stops the proliferation of T- and B-cells, and the immune system is suppressed (69,70).
Mycophenolate is generally prescribed at a fixed dose regardless of age and has been shown to effectively reduce the incidence of acute rejection and improve long-term graft survival when compared to azathioprine as an adjunct immunosuppressant therapy (69). Side effects are most commonly related to the gastrointestinal tract—mycophenolate frequently causes diarrhoea (30% patients), nausea, bloating, dyspepsia and vomiting (20% patients). More serious side effects include leukopenia, anaemia and thrombocytopenia (58).
Mycophenolate mofetil is rapidly absorbed in the upper gastrointestinal tract and hydrolyzed into MPA and hydroxyethyl morpholine (an inactive metabolite that is rapidly metabolized and then excreted in the urine) (42). Peak concentrations are achieved within 1–2 hours following oral administration. Enteric-coated mycophenolate sodium is more slowly absorbed, with peak drug concentrations occurring within 1.5–2.75 hours after administration (42). Mycophenolate mofetil and enteric-coated mycophenolic sodium formulations both have high oral bioavailability, approximately 80–90%. Drug distribution primarily occurs within the plasma compartment of the blood, with 97–99% of MPA bound to albumin. MPA metabolism is extensive and mostly occurs via the UGT system (71). The mostly inactive metabolite MPAG is then transported from liver cells into the bile. Once biliary excretion has occurred, MPA-7-O-glucuronide (MPAG) may be converted back into MPA by glucosidase produced by the intestinal flora, resulting in MPA reabsorption. This enterohepatic cycle of excretion and reabsorption can lead to a secondary peak within the concentration-time profile (42). Final elimination is by active tubular secretion of MPAG via the kidneys (72). MPA displays non-linear pharmacokinetics, with complex and large pharmacokinetic variability (73) partly attributed to patient serum albumin levels, renal function, genetics, drug-drug interactions (45,71,72,74).
Prednisolone and prednisone
Prednisolone and prednisone are corticosteroids that can interconvert. Prednisolone is the active drug moiety; prednisone is a pro-drug that undergoes reversible metabolism to the active metabolite (75). Corticosteroids are commonly included in initial immunosuppressant regimens at high doses to lower the risk of acute rejection (76), with doses tapered in the months following transplantation to lower doses (often <10 mg/day) (77). Prednisolone is usually given once daily in the morning to mimic the body’s natural diurnal rhythm of cortisol peak and trough concentrations (77).
Prednisolone blocks the immune system by depleting circulating T-cells through the inhibition of interleukin-2 signalling and production. Additionally, it inhibits transcription of the inflammatory cytokine interleukin-1, causing a secondary anti-inflammatory effect, which in turn, detrimentally effects wound healing (77,78). Side effects associated with long-term steroid use are common (54) and can include cosmetic changes (cushingoid appearance), growth retardation, osteoporosis, impaired wound healing, glucose intolerance and hyperlipidaemia (58,79). Although TDM is not necessary (80), it is crucial that doses be adjusted according to an individual’s response (managing/minimising side effects).
Studies examining immunosuppressant protocols in elderly recipients
Despite the possibility of physiological and metabolic changes with advancing age, few studies have examined the impact of a lower or altered immunosuppressant dosing protocol on graft outcomes in older adults. In 2009, Badowski and researchers lowered the immunosuppressant targets for a cohort of patients aged over 60 years. In this study, 101 subjects were allocated an initial regimen of mycophenolate mofetil 2 g/day and tacrolimus, target level 10–12 ng/mL; and 88 subjects 1 g/day and 8–10 ng/mL respectively (55). Dose adjustments were made when required, and overall, they found that a reduction in both tacrolimus target drug concentrations and maintenance mycophenolate dosing improved long-term graft survival, without a corresponding increase in the risk of acute rejection (P=0.006) (55). Despite these findings, there have been no follow-up studies at different clinics, and it remains largely unknown whether immunosuppressant doses or targets should be adjusted in elderly recipients.
Additionally, there is some contention about the inclusion of corticosteroids in immunosuppressive regimens in the elderly due to long-term toxicity (76). A review by Pascual et al. in 2012 showed that steroid withdrawal two weeks after transplantation was not associated with increased mortality or graft loss when patients were prescribed dual therapy with a CNI and mycophenolate (81). Le Meur et al. also concluded in 2015 that steroid avoidance or early withdrawal may improve patient outcomes in individuals with a low immunological risk (the elderly) (15). Resultantly, the US Registry’s current guidelines state that a post-transplant steroid-free regimen does not increase the risk of intermediate-term clinical outcomes (81), and a 2012 Annual Report in the USA showed that approximately 30% of all renal transplant recipients are prescribed steroid-free regimens (7). However, a more recent Cochrane review of steroid usage in adult kidney transplant recipients by Haller et al. in 2016 reported that steroid avoidance or withdrawal after transplantation increased the risk of acute rejection (82) and concluded that steroids should form part of maintenance immunosuppression (82). To date no clinical trials examining the impact of steroid withdrawal have been specifically done in elderly patients (45).
Studies examining immunosuppressant PK in elderly recipients
Only six studies have been explicitly designed to examine the pharmacokinetics of immunosuppressant medicines in elderly renal transplant recipients. Findings have been summarized in Table 1 and are critiqued below.
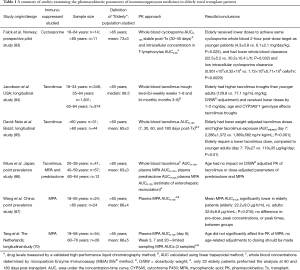
Full table
A prospective pilot study in 2008 by Falck et al. examined the pharmacokinetics of cyclosporine in 11 elderly and 14 younger adults. Elderly patients (>65 years) achieved similar 2-hour post-dose target levels as younger adults, with lower weight-adjusted doses (4.3±0.8 vs. 6.1±2.1 mg/kg/day; P=0.025). This led to the suggestion that drug clearance decreases with advancing age. Additionally, the intracellular concentration of cyclosporine in T-lymphocytes was measured, and it was found that the elderly had higher intracellular cyclosporine concentrations, despite similar whole blood concentrations. The authors suggested that in elderly patients a significantly larger proportion of the whole blood cyclosporine concentration may be located at the site of action (within the T-lymphocyte) indicating that in this population it might be safe to aim for an even lower target level than current guidelines (83).
In 2012, a retrospective, multi-centre study by Jacobson et al. examining 374 elderly renal transplant recipients over the first 6 months after transplantation, found that elderly patients (mean: 68.5 years) had higher dose-normalized tacrolimus trough concentrations than younger adults (84). After dose and body weight normalisation, tacrolimus concentrations were greater than 50% higher in elderly recipients, compared to younger adults (84). The study was retrospective, but involved large patient numbers (1,809 in total across all age groups). One study limitation was that only tacrolimus trough values were examined, rather than total drug exposure across the dosing interval.
A prospective study by David-Neto et al. [2017] involving 44 elderly patients and 31 younger adults examined tacrolimus exposure over time. Elderly recipients achieved higher target exposure and lower estimated total body clearance, with a lower normalized tacrolimus dose (85). The authors attribute this finding to a lower apparent clearance of tacrolimus in elderly patients. In this study, the average age of elderly patients was 65 years (85).
A prospective, point prevalence study conducted by Miura et al. in 2009 was the first to examine multiple immunosuppressant medicines in elderly patients, and showed that age had no impact on dose and weight adjusted pharmacokinetic parameters of tacrolimus, prednisolone and mycophenolate (Cmax, C0 and AUC0-12h) (86). However, only 12 of the 110 patients studied were elderly, which was defined as being 60–64 years of age (86). The study was undertaken at a single time-point, four weeks post-transplantation, hence, intra-patient variability and changes over time-post-transplantation were not considered.
Wang et al. [2007] were the first to examine the impact of aging on the pharmacokinetics of mycophenolate mofetil. MPA exposure, based on the area under the concentration-time curve (AUC), was significantly lower in elderly patients despite receiving the same MMF doses. There was however, no significant difference in peak drug concentration between the elderly and younger adult patient groups (87). This finding suggests that elderly patients have higher apparent clearance of mycophenolate, which contradicts our general understanding of the effects of aging on the pharmacokinetics of a drug. The authors instead suggest that elderly patients have more complex drug absorption processes (87). Overall, the sample size was relatively small, with 24 elderly and 24 younger adult patients examined. The mean age of the elderly patients was 65 years. As the study was a point-prevalence study, with each patients sampled between 10–12 weeks after transplantation, intra-patient variability could not be accounted for, which may explain the unusual finding.
A final study by Tang et al. in 2016, compared the effects of aging on the pharmacokinetics of mycophenolate in younger adults (54 patients) and the elderly (26 patients) (70). Patients were repetitively sampled over the first six months post-transplantation, with all patients given the same dose of mycophenolate mofetil. Researchers concluded that aging does not significantly alter mycophenolate exposure (AUC0–12h), which differs to the findings by Wang et al. (87) and Miura et al. (86). Authors suggested this may be due to the pharmacokinetics of mycophenolate being investigated repetitively both early and later post-transplantation. The mean age of the elderly recipients was 65 years (±4.9 years). Due to the necessary addition of tacrolimus as co-immunosuppressant therapy, the effects of ageing on tacrolimus were also examined. Tang et al.’s tacrolimus results mirrored David-Neto et al.’s (85) with both studies reporting that age significantly affects tacrolimus pharmacokinetics, with elderly patients having higher weight-normalized tacrolimus concentrations compared to younger adults (70).
Overall, these studies suggest that the doses of cyclosporine and tacrolimus required to achieve optimum immunosuppression in elderly patients may need to be lower than doses in younger adults, whilst mycophenolate doses should remain unchanged. No studies to date have looked at the effects of aging on MPA metabolites or free drug levels. Studies investigating the effects of aging on the pharmacokinetics of corticosteroids are also lacking. Unfortunately, few studies have included many patients over 70 years of age and hence it is still largely unknown how the physiological process of ageing affects the pharmacokinetic parameters of immunosuppressant medicines in this cohort (27).
Additionally, a number of population pharmacokinetic studies have been performed examining the pharmacokinetics of cyclosporine, tacrolimus and MPA in adult kidney transplant recipients [see Han 2013, Staatz 2011, Brooks 2016, and Kiang and Ensom, for reviews of these studies (65,66,71,74)]. These studies have not specifically examined the effect of being ‘elderly’ on the pharmacokinetics of immunosuppressant medicines, but rather looked at patient age in general, and as such, did not necessarily include a large number of elderly patients. Consequently, only some of these studies have identified patient age as an important covariate (88-92). As these studies generally look at multiple covariate factors, some of the effects of aging may also be explained by other patient factors, which can change with age (e.g., patient weight, renal function, haematocrit).
Studies examining immunosuppressant PD in elderly recipients
Studies examining immunosuppressant pharmacodynamics in elderly renal transplant recipients are lacking (Table 2). One study by Tang et al. [2016] examined baseline inosine 5’-monophosphate dehydrogenase (IMPDH) activity and MPA-induced IMPDH activity in 54 younger adults (mean age: 44 years) and 26 elderly (mean age: 65 years) renal transplant recipients. Patients were repetitively sampled for the first six months after transplantation. Overall, researchers found no difference in IMPDH activity pre-transplantation between the two age groups and there was no significant difference in AEC0–12h [area under the effect-time curve), EC50 (concentration that yields half the maximum response (31)] or Emax (70). This led researchers to conclude that age does not affect the pharmacodynamics of MPA (70). Clearly, further studies are required.

Full table
Transplant outcomes
Elderly renal transplant patients are at greatest risk of both morbidity and mortality (Table 3) (100). Elderly recipients are more susceptible to drug-related adverse effects, particular cardiovascular disease and infection (27,101), as well as more likely to suffer increased co-morbidity, both of which have been shown to increase post-transplant complications (27) and negatively impact patient survival (47,96).
Elderly recipients are more likely to die from infectious or cardiovascular-related causes due to side effects of immunosuppressant medicines, rather than graft failure (27,32,100). A study by Ojo et al. showed that renal transplant recipients aged over 65 years were seven times more likely to die with a functioning graft compared with young adults (aged 18–29 years) (46,100,102). This suggests that an optimal balance between immunosuppressant medicine efficacy and toxicity is not achieved in elderly recipients and further studies are needed to foster long-term graft and patient survival.
The risk of infection-related mortality has been shown to increase exponentially with age, due to reduced immune reactivity (29,103,104). Meier-Kriesche et al. reported in 2000 that elderly renal transplant recipients have a five-fold increase in their overall risk of death due to infection, compared with recipients aged between 30–39 years (105). Risk of infection, both opportunistic and non-opportunistic, is also increased with pharmacological over-immunosuppression, as infection is a side effect of immunosuppressant medicines (103). The literature suggests that lower maintenance immunosuppressant targets in elderly recipients may decrease patient susceptibility to side effects, which in turn, would lower the risk of infection (14), however, studies in patients aged above 70 years have yet to establish this (19,100,106).
Co-morbidities increase with advancing age, irrespective of transplantation. A study by Wu et al. [2005] examined the impact of patient co-morbidities on acute rejection and patient and graft survival in a cohort of 1,102 adult renal transplant patients (Table 3) (99). The study did not specifically examine elderly patients, however, 266 patients aged greater than 60 years (median age: 67 years) were included (99). Wu and researchers showed a trend toward an increasing number of co-morbidities over time, with significantly more patients having greater than five co-morbidities at the study’s conclusion (P<0.05). The results also showed that patients with greater than five co-morbidities were at significantly greater risk of death both immediately and more than three months post-transplantation, compared to patients with less than five co-morbidities (P<0.05) (99). With respect to age, researchers showed that elderly patients had the lowest survival rate, with survival decreasing with increasing age (P<0.05). Overall, this study serves to show the detrimental effect of co-morbidity number on both acute and long-term patient and graft outcomes (99).
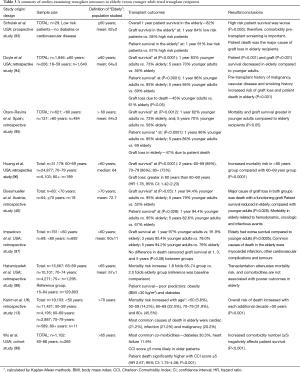
Full table
Conclusions
As the number of elderly patients undergoing renal transplantation continues to rise rapidly (107), it is crucial that current immunosuppressant prescribing practices be more clearly examined and defined. It is imperative that the effects of aging on the pharmacokinetics and pharmacodynamics of immunosuppressant medicines be established so that the most safe and efficacious immunosuppressive regimens are prescribed.
From the limited studies done to date, it appears that elderly transplant recipients have higher tacrolimus exposure (up to 50% greater), despite receiving lower adjusted doses, whilst mycophenolate exposure remains unchanged. These preliminary findings support the assumption that CYP associated metabolism is more likely to be affected by aging than metabolism by UGTs (108). Future studies should examine MPAG exposure in the elderly, as this is likely to increase with reduced patient renal function. MPAG competes with MPA for protein binding sites, and a reduction in renal function may increase MPAG levels in the elderly. Resultantly, higher MPAG levels could potentially influencing mycophenolate dosing, if adjustments were made according to total drug exposure. Further studies are also required to ascertain the effect of ageing on intracellular drug levels in lymphocytes.
Based on the results of a single study, there appears to be no difference in the effect of mycophenolate on IMPDH activity between elderly and younger adult recipients, however, further studies are required to corroborate and confirm this finding. Future studies should also examine CNIs, in terms of their maximum effect (Emax) on calcineurin inhibition, as currently, elderly recipients are most likely to suffer CNI side effects, but it is not completely understood as to why. What is clear is that the elderly are at greatest risk of immunosuppressant side effects, with the three main causes of death being infection, malignancy and cardiovascular disease attributed to immunosuppressant medicine usage (27,45,100,101). This suggests that the pharmacodynamic effects of immunosuppressant medicines are influenced by age. It is difficult to know with certainty whether a difference in baseline reserve or difference in drug sensitivity (31) (or both) is responsible for the change in patient responsiveness to immunosuppressant medicines in elderly renal transplant recipients.
Like any transplant group, the elderly are a heterogeneous sub-population and this influences patient responsiveness to drugs. Because of this unpredictability, elderly renal transplant recipients may not only require less immunosuppression but also specific, individualised changes, as immunosenescence affects some immunological targets more so than others (45). The complexity surrounding appropriate immunosuppressant dosing is compounded by the usage of combination immunosuppressant drug treatments. Currently, it is unknown whether elderly recipients justly require the same level of immunosuppression to support graft outcomes as younger adults, nor which immunosuppressant agents or target concentrations best achieve optimal patient and graft outcomes (106,109). Further studies are required to characterise these age-related changes (110).
Acknowledgments
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Hod T, Goldfarb-Rumyantzev AS. Clinical issues in renal transplantation in the elderly. Clin Transplant 2015;29:167-75. [Crossref] [PubMed]
- Oniscu GC, Brown H, Forsythe JL. How great is the survival advantage of transplantation over dialysis in elderly patients? Nephrol Dial Transplant 2004;19:945-51. [Crossref] [PubMed]
- Wolfe RA, Ashby VB, Milford EL, et al. Comparison of mortality in all patients on dialysis, patients on dialysis awaiting transplantation, and recipients of a first cadaveric transplant. N Engl J Med 1999;341:1725-30. [Crossref] [PubMed]
- Cooper M, Forland CL. The elderly as recipients of living donor kidneys, how old is too old? Curr Opin Organ Transplant 2011;16:250-5. [Crossref] [PubMed]
- Huang E, Segev DL, Rabb H. Kidney transplantation in the elderly. Semin Nephrol 2009;29:621-35. [Crossref] [PubMed]
- Segall L, Nistor I, Pascual J, et al. Criteria for and Appropriateness of Renal Transplantation in Elderly Patients With End-Stage Renal Disease: A Literature Review and Position Statement on Behalf of the European Renal Association-European Dialysis and Transplant Association Descartes Working Group and European Renal Best Practice. Transplantation 2016;100:e55-65. [Crossref] [PubMed]
- U.S. Department of Health and Human Services. OPTN/SRTR 2012 Annual Data Report: Kidney 2012 Annual Data Report. USA, 2012:11-44.
- ANZDATA Registry. The 39th Annual ANZDATA Report Chapter 8: Transplantation 2016.
- Pippias M, Stel VS, Abad Diez JM, et al. Renal replacement therapy in Europe: a summary of the 2012 ERA-EDTA Registry Annual Report. Clin Kidney J 2015;8:248-61. [Crossref] [PubMed]
- Maggiore U, Abramowicz D, Budde K. Renal transplantation in the elderly. Transplant Rev (Orlando) 2015;29:191-2. [Crossref] [PubMed]
- Morris P, Knechtle SJ. Results of Renal Transplantation. In: Patzer RE, Knechtle SJ. editors. Kidney Transplantation - Principles and Practice. UK: Elsevier, 2014:676-97.
- Aubert O, Kamar N, Vernerey D, et al. Long term outcomes of transplantation using kidneys from expanded criteria donors: prospective, population based cohort study. BMJ 2015;351:h3557. [Crossref] [PubMed]
- Karim A, Farrugia D, Cheshire J, et al. Recipient age and risk for mortality after kidney transplantation in England. Transplantation 2014;97:832-8. [Crossref] [PubMed]
- Macrae J, Friedman AL, Friedman EA, et al. Live and deceased donor kidney transplantation in patients aged 75 years and older in the United States. Int Urol Nephrol 2005;37:641-8. [Crossref] [PubMed]
- Le Meur Y. What immunosuppression should be used for old-to-old recipients? Transplant Rev (Orlando) 2015;29:231-6. [Crossref] [PubMed]
- Ponticelli C, Podesta MA, Graziani G. Renal transplantation in elderly patients. How to select the candidates to the waiting list? Transplant Rev (Orlando) 2014;28:188-92. [Crossref] [PubMed]
- Rao PS, Merion RM, Ashby VB, et al. Renal transplantation in elderly patients older than 70 years of age: results from the Scientific Registry of Transplant Recipients. Transplantation 2007;83:1069-74. [Crossref] [PubMed]
- Frei U, Noeldeke J, Machold-Fabrizii V, et al. Prospective age-matching in elderly kidney transplant recipients--a 5-year analysis of the Eurotransplant Senior Program. Am J Transplant 2008;8:50-7. [PubMed]
- Sutherland AI, IJzermans JN, Forsythe JL, et al. Kidney and liver transplantation in the elderly. Br J Surg 2016;103:e62-72. [Crossref] [PubMed]
- Heinbokel T, Elkhal A, Liu G, et al. Immunosenescence and organ transplantation. Transplant Rev (Orlando) 2013;27:65-75. [Crossref] [PubMed]
- Klinger M, Banasik M. Immunological characteristics of the elderly allograft recipient. Transplant Rev (Orlando) 2015;29:219-23. [Crossref] [PubMed]
- Abecassis M, Bridges ND, Clancy CJ, et al. Solid-organ transplantation in older adults: current status and future research. Am J Transplant 2012;12:2608-22. [Crossref] [PubMed]
- McLachlan AJ, Hilmer SN, Le Couteur DG. Variability in response to medicines in older people: phenotypic and genotypic factors. Clin Pharmacol Ther 2009;85:431-3. [Crossref] [PubMed]
- de Oliveira Baldoni A, Drumond Chequer FM, Ferraz ERA, et al. Elderly and drugs: risks and necessity of rational use. Brazilian Journal of Pharmaceutical Sciences 2010;46:617-32. [Crossref]
- Hutchison LC, O'Brien CE. Changes in Pharmacokinetics and Pharmacodynamics in the Elderly Patient. Journal of Pharmacy Practice 2016;20:4-12. [Crossref]
- Pascual J, Zamora J, Pirsch JD. A systematic review of kidney transplantation from expanded criteria donors. Am J Kidney Dis 2008;52:553-86. [Crossref] [PubMed]
- Shi YY, Hesselink DA, van Gelder T. Pharmacokinetics and pharmacodynamics of immunosuppressive drugs in elderly kidney transplant recipients. Transplant Rev (Orlando) 2015;29:224-30. [Crossref] [PubMed]
- Johnson DW, Herzig K, Purdie D, et al. A comparison of the effects of dialysis and renal transplantation on the survival of older uremic patients. Transplantation 2000;69:794-9. [Crossref] [PubMed]
- Meier-Kriesche HU, Ojo AO, Hanson JA, et al. Exponentially increased risk of infectious death in older renal transplant recipients. Kidney Int 2001;59:1539-43. [Crossref] [PubMed]
- Lim WH, Dogra G, Chadban SJ, et al. Lack of impact of donor age on patient survival for renal transplant recipients ≥60years. Transpl Int 2012;25:401-8. [Crossref] [PubMed]
- Jacobson S. Effects of Pharmacokinetic and Pharmacodynamic Changes in the Elderly. Psychiatry Times 2013:26-9.
- Turnheim K. When drug therapy gets old: pharmacokinetics and pharmacodynamics in the elderly. Experimental Gerontology 2003;38:843-53. [Crossref] [PubMed]
- Aymanns C, Keller F, Maus S, et al. Review on pharmacokinetics and pharmacodynamics and the aging kidney. Clin J Am Soc Nephrol 2010;5:314-27. [Crossref] [PubMed]
- Colloca G, Santoro M, Gambassi G. Age-related physiologic changes and perioperative management of elderly patients. Surg Oncol 2010;19:124-30. [Crossref] [PubMed]
- Birkett DJ. Pharmacokinetics Made Easy 2nd ed. pocket guide ed. Australia: McGraw Hill Education, 2010.
- Petzinger E, Geyer J. Drug transporters in pharmacokinetics. Naunyn Schmiedebergs Arch Pharmacol 2006;372:465-75. [Crossref] [PubMed]
- Stieger B, Hagenbuch B. Recent advances in understanding hepatic drug transport. F1000 Research 2016;5:2465. [Crossref] [PubMed]
- van Assema DM, Lubberink M, Boellaard R, et al. P-glycoprotein function at the blood-brain barrier: effects of age and gender. Mol Imaging Biol 2012;14:771-6. [Crossref] [PubMed]
- McLean AJ, Le Couteur DG. Aging biology and geriatric clinical pharmacology. Pharmacol Rev 2004;56:163-84. [Crossref] [PubMed]
- Corsonello A, Pedone C, Antonelli Incalzi R. Age-Related Pharmacokinetic and Pharmacodynamic Related Risk of Adverse Drug Reactions. Current Medicinal Chemistry 2010;17:571-84. [Crossref] [PubMed]
- Hämmerlein A, Derendorf H, Lowenthal DT. Pharmacokinetic and Pharmacodynamic Changes in the Elderly: Clinical Implications. Clin Pharmacokinet 1998;35:49-64. [Crossref] [PubMed]
- Milone MC. Chapter 1 - Overview of the pharmacology and toxicology of immunosuppressant agents that require therapeutic drug monitoring. Personalized Immunosuppression in Transplantation 2016.
- Albright JF, Albright JW. Aging of Adaptive/Acquired Immunity. Aging, Immunity, and Infection. Totowa, NJ: Humana Press, 2003:135-211.
- Gibson KL, Wu YC, Barnett Y, et al. B-cell diversity decreases in old age and is correlated with poor health status. Aging Cell 2009;8:18-25. [Crossref] [PubMed]
- Krenzien F, ElKhal A, Quante M, et al. A Rationale for Age-Adapted Immunosuppression in Organ Transplantation. Transplantation 2015;99:2258-68. [Crossref] [PubMed]
- Boesmueller C, Biebl M, Scheidl S, et al. Long-term outcome in kidney transplant recipients over 70 years in the Eurotransplant Senior Kidney Transplant Program: a single center experience. Transplantation 2011;92:210-6. [Crossref] [PubMed]
- Peeters LEJ, Andrews LM, Hesselink DA, et al. Personalized immunosuppression in elderly renal transplant recipients. Pharmacol Res 2018;130:303-7. [Crossref] [PubMed]
- Kuypers DRJ. Immunotherapy in Elderly Transplant Recipients: A Guide to Clinically Significant Drug Interactions. Drugs Aging 2009;26:715-37. [Crossref] [PubMed]
- Seyda M, Quante M, Uehara H, et al. Immunosenescence in renal transplantation: a changing balance of innate and adaptive immunity. Curr Opin Organ Transplant 2015;20:417-23. [Crossref] [PubMed]
- Halleck F, Khadzhynov D, Liefeldt L, et al. Immunologic outcome in elderly kidney transplant recipients: is it time for HLA-DR matching? Nephrol Dial Transplant 2016;31:2143-9. [Crossref] [PubMed]
- Barraclough KA, Staatz CE, Johnson DW, et al. Kidney transplant outcomes are related to tacrolimus, mycophenolic acid and prednisolone exposure in the first week. Transpl Int 2012;25:1182-93. [Crossref] [PubMed]
- Chadban SJ, Barraclough KA, Campbell SB, et al. KHA-CARI guideline: KHA-CARI adaptation of the KDIGO Clinical Practice Guideline for the Care of Kidney Transplant Recipients. Nephrology (Carlton) 2012;17:204-14. [Crossref] [PubMed]
- Renal transplantation and the older adult patient [Internet]. Wolters Kluwer Health 2016 [cited 30/11/2017]. Available online: https://www.uptodate.com/contents/renal-transplantation-and-the-older-adult-patient#H1
- Chadban SJ, Barraclough KA, Campbell SB, et al. The Kidney Health Australia-Caring for Australasians with Renal Impairment (KHA-CARI) Guidelines. Nephrology 2012;17:204-14. [Crossref] [PubMed]
- Badowski M, Gurk-Turner C, Cangro C, et al. The impact of reduced immunosuppression on graft outcomes in elderly renal transplant recipients. Clin Transplant 2009;23:930-7. [Crossref] [PubMed]
- Karpe KM, Talaulikar GS, Walters GD. Calcineurin inhibitor withdrawal or tapering for kidney transplant recipients. Cochrane Database Syst Rev 2017;7:CD006750. [PubMed]
- Morris P, Knechtle SJ. Calcineurin Inhibitors. In: Mejia JC, Basu A, Shapiro R. editors. Kidney Transplantation - Principles and Practice. UK: Elsevier, 2014:231-49.
- Muntean A, Lucan M. Immunosuppression in Kidney Transplantation. Clujul Medical 2013;86:177-80. [PubMed]
- Kim IW, Moon YJ, Ji E, et al. Clinical and genetic factors affecting tacrolimus trough levels and drug-related outcomes in Korean kidney transplant recipients. Eur J Clin Pharmacol 2012;68:657-69. [Crossref] [PubMed]
- Okada A, Ushigome H, Kanamori M, et al. Population pharmacokinetics of cyclosporine A in Japanese renal transplant patients: comprehensive analysis in a single center. Eur J Clin Pharmacol 2017;73:1111-9. [Crossref] [PubMed]
- Lloberas N, Elens L, Llaudo I, et al. The combination of CYP3A4*22 and CYP3A5*3 single-nucleotide polymorphisms determines tacrolimus dose requirement after kidney transplantation. Pharmacogenet Genomics 2017;27:313-22. [Crossref] [PubMed]
- Andreu F, Colom H, Elens L, et al. A New CYP3A5*3 and CYP3A4*22 Cluster Influencing Tacrolimus Target Concentrations: A Population Approach. Clin Pharmacokinet 2017;56:963-75. [Crossref] [PubMed]
- Staatz CE, Tett SE. Pharmacokinetic Considerations Relating to Tacrolimus Dosing in the Elderly. Drugs Aging 2005;22:541-57. [Crossref] [PubMed]
- Staatz CE, Willis C, Taylor PJ, et al. Population pharmacokinetics of tacrolimus in adult kidney transplant recipients. Clin Pharmacol Ther 2002;72:660-9. [Crossref] [PubMed]
- Brooks E, Tett SE, Isbel NM, et al. Population Pharmacokinetic Modelling and Bayesian Estimation of Tacrolimus Exposure: Is this Clinically Useful for Dosage Prediction Yet? Clin Pharmacokinet 2016;55:1295-335. [Crossref] [PubMed]
- Han K, Pillai VC, Venkataramanan R. Population pharmacokinetics of cyclosporine in transplant recipients. AAPS J 2013;15:901-12. [Crossref] [PubMed]
- McKay DB, Steinberg SM. Kidney Transplantation: A Guide to the Care of Kidney Transplant Recipients. New York: Springer, 2010.
- Johnson DW, Nicol DL, Purdie DM, et al. Is Mycophenolate Mofetil less safe than Azathioprine in Elderly Renal Transplant Recipients? Transplantation 2002;73:1158-63. [Crossref] [PubMed]
- Salvadori M, Holzer H, Mattos AD, et al. Enteric-Coated Mycophenolate Sodium is Therapeutically Equivalent to Mycophenolate Mofetil inde novoRenal Transplant Patients. American Journal of Transplantation 2004;4:231-6. [Crossref] [PubMed]
- Tang JT, de Winter BC, Hesselink DA, et al. The pharmacokinetics and pharmacodynamics of mycophenolate mofetil in younger and elderly renal transplant recipients. Br J Clin Pharmacol 2017;83:812-22. [Crossref] [PubMed]
- Kiang TKL, Ensom MHH. Population Pharmacokinetics of Mycophenolic Acid: An Update. Clin Pharmacokinet 2018;57:547-58. [Crossref] [PubMed]
- Božina N, Lalic Z, Nad-Skegro S, et al. Steady-state pharmacokinetics of mycophenolic acid in renal transplant patients: exploratory analysis of the effects of cyclosporine, recipients' and donors' ABCC2 gene variants, and their interactions. Eur J Clin Pharmacol 2017;73:1129-40. [Crossref] [PubMed]
- Staatz CE, Duffull SB, Kiberd B, et al. Population pharmacokinetics of mycophenolic acid during the first week after renal transplantation. Eur J Clin Pharmacol 2005;61:507-16. [Crossref] [PubMed]
- Staatz CE, Tett SE, Maximum A. Posteriori Bayesian Estimation of Mycophenolic Acid Area Under the Concentration-Time Curve: Is This Clinically Useful for Dosage Prediction Yet? Clin Pharmacokinet 2011;50:759-72. [Crossref] [PubMed]
- Bergmann TK, Barraclough KA, Lee KJ, et al. Clinical pharmacokinetics and pharmacodynamics of prednisolone and prednisone in solid organ transplantation. Clin Pharmacokinet 2012;51:711-41. [Crossref] [PubMed]
- Haller MC, Kammer M, Kainz A, et al. Steroid withdrawal after renal transplantation: a retrospective cohort study. BMC Med 2017;15:8. [Crossref] [PubMed]
- Morris P, Knechtle SJ. Steroids. In: Knight SR, editor. Kidney Transplantation – Principles and Practice. 7 ed. UK: Elsevier, 2014:221-30.
- Glucocorticoid effects on the immune system [Internet]. Wolters Kluwer Health 2017. Available online: https://www.uptodate.com/contents/glucocorticoid-effects-on-the-immune-system
- Morton JM, Williamson S, Kear LM, et al. Therapeutic drug monitoring of prednisolone after lung transplantation. J Heart Lung Transplant 2006;25:557-63. [Crossref] [PubMed]
- Potter JM, McWhinney BC, Sampson L, et al. Area-Under-the-Curve Monitoring of Prednisolone for Dose Optimization in a Stable Renal Transplant Population. Ther Drug Monit 2004;26:408-14. [Crossref] [PubMed]
- Pascual J, Royuela A, Galeano C, et al. Very early steroid withdrawal or complete avoidance for kidney transplant recipients: a systematic review. Nephrol Dial Transplant 2012;27:825-32. [Crossref] [PubMed]
- Haller MC, Royuela A, Nagler EV, et al. Steroid avoidance or withdrawal for kidney transplant recipients. Cochrane Database Syst Rev 2016.CD005632. [PubMed]
- Falck P, Asberg A, Byberg KT, et al. Reduced elimination of cyclosporine A in elderly (>65 years) kidney transplant recipients. Transplantation 2008;86:1379-83. [Crossref] [PubMed]
- Jacobson PA, Schladt D, Oetting WS, et al. Lower calcineurin inhibitor doses in older compared to younger kidney transplant recipients yield similar troughs. Am J Transplant 2012;12:3326-36. [Crossref] [PubMed]
- David-Neto E, Romano P, Kamada Triboni AH, et al. Longitudinal Pharmacokinetics of Tacrolimus in Elderly Compared With Younger Recipients in the First 6 Months After Renal Transplantation. Transplantation 2017;101:1365-72. [Crossref] [PubMed]
- Miura M, Satoh S, Kagaya H, et al. No impact of age on dose-adjusted pharmacokinetics of tacrolimus, mycophenolic acid and prednisolone 1 month after renal transplantation. Eur J Clin Pharmacol 2009;65:1047-53. [Crossref] [PubMed]
- Wang CX, Meng FH, Chen LZ, et al. Population pharmacokinetics of mycophenolic acid in senile Chinese kidney transplant recipients. Transplant Proc 2007;39:1392-5. [Crossref] [PubMed]
- Falck P, Midtvedt K, Vân Lê TT, et al. A Population Pharmacokinetic Model of Ciclosporin Applicable for Assisting Dose Management of Kidney Transplant Recipients. Clin Pharmacokinet 2009;48:615-23. [Crossref] [PubMed]
- Wu KH, Cui YM, Guo JF, et al. Population pharmacokinetics of cyclosporine in clinical renal transplant patients. Drug Metab Dispos 2005;33:1268-75. [Crossref] [PubMed]
- Ogasawara K, Chitnis SD, Gohh RY, et al. Multidrug resistance-associated protein 2 (MRP2/ABCC2) haplotypes significantly affect the pharmacokinetics of tacrolimus in kidney transplant recipients. Clin Pharmacokinet 2013;52:751-62. [Crossref] [PubMed]
- Størset E, Holford N, Midtvedt K, et al. Importance of hematocrit for a tacrolimus target concentration strategy. Eur J Clin Pharmacol 2014;70:65-77. [Crossref] [PubMed]
- Veličković-Radovanović RM, Jankovic SM, Milovanovic JR, et al. Variability of mycophenolic acid elimination in the renal transplant recipients - population pharmacokinetic approach. Ren Fail 2015;37:652-8. [Crossref] [PubMed]
- Schulak JA, Hricik DE. Kidney transplantation in the elderly. Geriatr Nephrol Urol 1991;1:105-12. [Crossref]
- Doyle SE, Matas AJ, Gillingham K, et al. Predicting clinical outcome in the elderly renal transplant recipient. Kidney Int 2000;57:2144-50. [Crossref] [PubMed]
- Otero-Raviña F, Rodriguez-Martinez M, Gude F, et al. Renal transplantation in the elderly: does patient age determine the results? Age Ageing 2005;34:583-7. [Crossref] [PubMed]
- Huang E, Poommipanit N, Sampaio MS, et al. Intermediate-term outcomes associated with kidney transplantation in recipients 80 years and older: an analysis of the OPTN/UNOS database. Transplantation 2010;90:974-9. [Crossref] [PubMed]
- Impedovo SV, Ditonno P, Ricapito V, et al. Advanced age is not an exclusion criterion for kidney transplantation. Transplant Proc 2013;45:2650-3. [Crossref] [PubMed]
- Hatamizadeh P, Molnar MZ, Streja E, et al. Recipient-related predictors of kidney transplantation outcomes in the elderly. Clin Transplant 2013;27:436-43. [Crossref] [PubMed]
- Wu C, Evans I, Joseph R, et al. Comorbid conditions in kidney transplantation: association with graft and patient survival. J Am Soc Nephrol 2005;16:3437-44. [Crossref] [PubMed]
- Dreyer GJ, Hemke AC, Reinders ME, et al. Transplanting the elderly: Balancing aging with histocompatibility. Transplant Rev (Orlando) 2015;29:205-11. [Crossref] [PubMed]
- Cameron JS. Renal transplantation in the elderly. Int Urol Nephrol 2000;32:193-201. [Crossref] [PubMed]
- Ojo AO, Hanson JA, Wolfe RA, et al. Long-term survival in renal transplant recipients with graft function. Kidney Int 2000;57:307-13. [Crossref] [PubMed]
- Lehner LJ, Staeck O, Halleck F, et al. Need for optimized immunosuppression in elderly kidney transplant recipients. Transplant Rev (Orlando) 2015;29:237-9. [Crossref] [PubMed]
- Welzl K, Kern G, Mayer G, et al. Effect of different immunosuppressive drugs on immune cells from young and old healthy persons. Gerontology 2014;60:229-38. [Crossref] [PubMed]
- Meier-Kriesche HU, Ojo A, Hanson J, et al. Increased Immunosuppressive Vulnerability in Elderly Renal Transplant Recipients. Transplantation 2000;69:885-9. [Crossref] [PubMed]
- Danovitch GM, Gill J, Bunnapradist S. Immunosuppression of the elderly kidney transplant recipient. Transplantation 2007;84:285-91. [Crossref] [PubMed]
- ANZDATA Registry. The 39th Annual ANZDATA Report Chapter 2: Prevalence of End Stage Kidney Disease 2016.
- McLachlan AJ, Pont LG. Drug metabolism in older people--a key consideration in achieving optimal outcomes with medicines. J Gerontol A Biol Sci Med Sci 2012;67:175-80. [Crossref] [PubMed]
- Andrés A, Budde K, Clavien PA, et al. A randomized trial comparing renal function in older kidney transplant patients following delayed versus immediate tacrolimus administration. Transplantation 2009;88:1101-8. [Crossref] [PubMed]
- Staatz CE, Goodman LK, Tett SE. Effect of CYP3A and ABCB1 Single Nucleotide Polymorphisms on the Pharmacokinetics and Pharmacodynamics of Calcineurin Inhibitors: Part II. Clin Pharmacokinet 2010;49:207-21. [Crossref] [PubMed]


