Testicular versus percutaneous epididymal sperm aspiration for patients with obstructive azoospermia: a systematic review and meta-analysis
Introduction
Obstructive azoospermia (OA) is one of the major causes of infertility in men (1), and is particularly frequent in men with vasectomy, reproductive tract trauma, or congenital absence of the vas deferens but normal spermatogenesis. The clinical management of OA depends on etiology but usually includes either (I) surgery to resolve obstruction or (II) sperm retrieval for intracytoplasmic sperm injection (ICSI) (1). Although the former method permits natural fertilization, the reproductive tract may be difficult to approach or reconstruct in some cases. Thus, ICSI may be the more feasible solution for these patients. First applied in 1992 (2), ICSI has evolved to become a reliable therapy for the majority of male reproductive tract deficiencies (3). However, numerous paternal and maternal factors can influence ICSI outcome. A major paternal factor is the source of sperm. Percutaneous epididymal sperm aspiration (PESA) and testicular sperm aspiration (TESA) are two common sperm retrieval approaches for ICSI in men with severe OA (1), but the more appropriate choice for sperm retrieval is still debated.
Several studies have directly compared ICSI outcomes between PESA and TESA treatment groups but results have not been unanimous. A retrospective study from a private fertility center in Brazil found no significant difference in pregnancy rate between these two procedures but did report non-similarity in other outcomes (4). Another large-scale retrospective study from Egypt also reported that the source of sperm used for ICSI treatment of OA did not affect fertilization, pregnancy, or miscarriage rates (5). Alternatively, one report found a higher fertilization rate in the PESA group among OA cases (6). Despite debate for more than 15 years, whether PESA or TESA yields better ICSI outcomes for OA remains unresolved. Some of these studies included patients with non-OA (NOA) and OA in the same group; however, the etiologies of NOA and OA are entirely different (7), so it is more reasonable to compare ICSI outcomes separately for these two types of azoospermia. Further, these data were gathered from single centers, which may the applicability of the results to the general OA population. Hence, our study aimed to compare ICSI outcomes between PESA and TESA among patients with OA across centers by meta-analysis.
Methods
Our systematic review and meta-analysis followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines (8). This synthesized study was exempted from institutional review board approval because it used published data. An experienced researcher (YN Kang) and a urologist (CC Wu) coordinated the study (9-11).
Inclusion and exclusion criteria
Study eligibility criteria were established prior to literature search and selection. The inclusion criteria were (I) men with OA, (II) undergoing ICSI, and (III) comparing TESA to PESA. The exclusion criteria were as follows: (I) mixed data of OA with other conditions without stratified analysis; (II) book contents, meeting reports, or news articles; (III) gray literature without details; (IV) no report of relevant outcomes (fertilization rate, implantation rate, pregnancy rate, miscarriage rate, complications/adverse events). Two reviewers (YN Kang and PY Shen) individually identified the relevant studies according to the criteria. Disagreements were resolved through discussion with a third author (CC Wu).
Data sources and search strategy
Four electronic databases were searched for potential references: Embase, PubMed (including MEDLINE), ScienceDirect, and Web of Science. The primary search strategy was established in PubMed using common terms, medical subject headings, and abbreviations related to OA and ICSI. Synonyms were combined by the Boolean operator “OR” and the concepts of OA and ICSI were combined using the Boolean operator “AND”. The search strategy included no restrictions on publication date or language to include studies from countries across the globe. The final searches were completed before 28 June 2019 (Supplementary file 1).
Evidence selection
After potential references were identified, two investigators (PY Shen and YN Kang) independently selected eligible studies in two steps. First, the title and abstract were screened for relevance. Articles appearing relevant according to title and abstract review were then subjected to full review. The two investigators excluded articles unrelated to OA, ICSI, and TESA according to eligible criteria, and then retrieved full-texts of the remaining articles for further review. The two investigators removed ineligible studies without details on patient condition or predefined study endpoints. A senior urologist made the final judgment in case of disagreement.
Data extraction
Two investigators (PY Shen and YN Kang) independently extracted the following information and outcome data from the included studies: first author, publication year, study design, number of patients, study period, paternal mean age, maternal mean age, mean number of ICSI cycles, and outcomes (including fertilization rate, implantation rate, pregnancy rate, and miscarriage rate). The outcomes were usually reported as event rates. If the original report presented outcomes as percentages, they were converted to event rates. If the original report only showed mean ± standard deviation, we cannot convert in event rates because of no denominator.
Quality assessment
Most of the included studies were retrospective, so two authors (PY Shen and KW Shih) independently appraised study quality using the Newcastle-Ottawa Quality Assessment Scale. When a given study was rated differently, a third investigator (YN Kang) critically reviewed risk of bias for the disagreement and made the final rating.
Statistical analysis
We used a pairwise meta-analysis for statistical synthesis. Odds ratio (OR) was chosen to express pooled results because the outcomes were binary data and most studies were retrospective. To overcome the variability among study populations and treatments, quantitative syntheses were conducted using a random-effects model. Moreover, to compare rare events, the Mantel-Haenszel method was employed. The results of this meta-analysis are presented as forest plots including events, total numbers, and ORs with [95% confidence intervals (CIs)].
According to the Cochrane Handbook for Systematic Reviews of Interventions, a pooled result may be influenced by heterogeneity across studies and by publication bias. Thus, heterogeneity and small study effects should be examined (12). Usually, heterogeneity is tested and presented as tau-square and I-square (I2) values, so we calculated these two values and judged heterogeneity of pooled results according to I2 and P values for tau-square. I2, the most common statistics for heterogeneity across studies, represents the proportion of total variation among studies due to heterogeneity (with I2>75% considered highly heterogeneous). Pooled results were also deemed highly heterogeneous when the P value for tau-square was lower than 0.10 (12). Funnel plots were constructed and assessed by Egger’s test for publication bias. Pooled results were considered biased by publication when Egger’s test reached statistical significance.
We also conducted subgroup analyses according to paternal age group for all outcomes because paternal age is an important factor determining ICSI success. According to a previous study (13), paternal age was stratified into three subgroups: <30 years old, 30–35 years old, and >35 years old. In addition, maternal age is an important factor in human reproduction, so we also conducted subgroup analyses according to maternal mean age. Based on available data and previous work (13,14), we used <30 years old, 30–35 years old, and >35 years old for subgroup stratification. To clarify the effect of sperm aspiration technique on miscarriage, we further stratified data by number of ICSI cycles because there appears to be a cumulative increase in risk (14).
Pairwise meta-analyzes were completed in Review Manager (version 5.3, The Cochrane Collaboration, Oxford, UK), and the small study effects were detected using Comprehensive Meta-Analysis (version 2) for Microsoft Windows 10.
Results
Literature search results
The initial literature search retrieved a total of 2,965 references from Embase (n=1,097), PubMed (n=715), ScienceDirect (n=344), and Web of Science (n=809). Of these, 1,018 duplicates were removed, and 1,934 were excluded as unrelated documents (n=37), for not enrolling OA patients (n=1,314), for not using ICSI (n=119), for having no PESA treatment group (n=428), or having no TESA treatment group (n=36) according to title and abstract screening. Full-text review of the remaining 13 references excluded five publications, one review, two studies without comparison of PESA versus TESA, and two gray literature documents without the required details (15,16). Finally, seven retrospective studies and one prospective study were included in the qualitative and quantitative synthesis (5,6,13,17-21). Figure 1 shows the process of study selection for this comprehensive review.
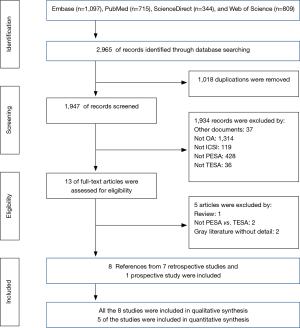
Flow diagram of evidence selection according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. OA, obstructive azoospermia; ICSI, intracytoplasmic sperm injection; TESA, testicular sperm aspiration; PESA, percutaneous epididymal sperm aspiration.
Overview of the included studies
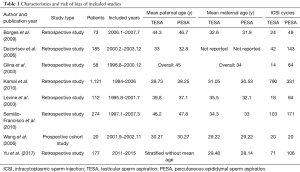
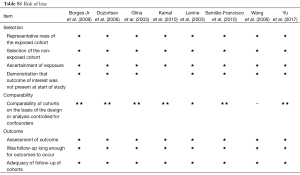
The included studies involved 2,020 men receiving 2,060 ICSI cycles. Table 1 summarizes basic study characteristics including first author surname, publication year, study design, number of patients, study period, paternal mean age, maternal mean age, and mean number of ICSI cycles. These studies covered the period from the early 1990s to 2015 and thus included those from the early years of TESA and PESA application to near present day. The studies were conducted in Brazil (6,17,18,20), China (13,21), Egypt (5), and the USA (19). According to the available information, paternal mean age ranged from 30 to 47 years and maternal mean age from 28 to 35 years. Seven of the 8 included studies provided adequate information on study design, although results may still be influenced by bias due to differences in clinical processes. The other (retrospective) study presented limited information on methodology and bias control (21). Complete information on quality assessment is presented in Table S1.
Full table
Full table
Fertilization
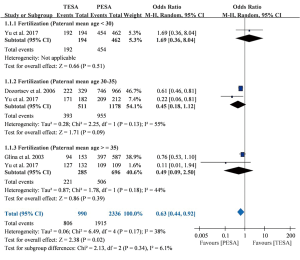
Three of the eight studies, including 420 men receiving 440 ICSI cycles, reported data on fertilization rate (6,13,18). The pooled estimate indicated that TESA led to significantly lower fertilization rate than PESA (OR =0.63, 95% CI: 0.44–0.92, P=0.02) with moderate heterogeneity among studies (I2=38%; P=0.17) (Figure S1). However, subgroup analysis did not reveal significant differences in fertilization rates between TESA and PESA treatment arms, although there was a favorable trend for greater PESA success among the subgroup with mean paternal age from 30 to 35 years old. In this subgroup, TESA yielded a lower fertilization rate than PESA but without statistical significance (OR =0.45, 95% CI: 0.18–1.12, P=0.09). There was also substantial heterogeneity among studies (I2=55%; P=0.13).
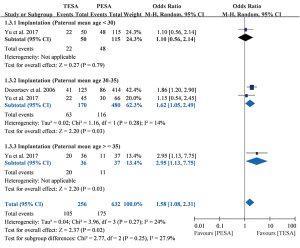
Implantation
Two studies including 362 patients receiving 362 ICSI cycles reported implantation rates (6,13). Pooled results showed that TESA yielded a significantly higher implantation rate than PESA (OR =1.58, 95% CI: 1.08–2.31, P=0.02) and heterogeneity was low (I2=24%; P=0.27) (Figure 2). In one of these studies (13), significantly higher implantation rates were found in the TESA subgroups 30–35 years (OR =1.62, 95% CI: 1.05–2.49, P=0.03) and older than 35 years (OR =2.95, 95% CI: 1.13–7.75, P=0.03), but not in the younger than 30 years subgroup (OR =1.10, 95% CI: 0.56–2.14, P=0.79).
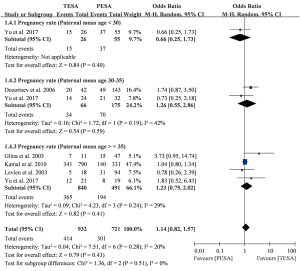
Pregnancy
A total of five studies including 1,541 men receiving 1,673 ICSI cycles presented data on pregnancy rates (5,6,13,18,19). The pooled results revealed no significant difference between TESA and PESA (OR =1.14, 95% CI: 0.82–1.57, P=0.43) and heterogeneity was low (I2=20%; P=0.28) (Figure 3). Similar trends were also found in subgroups of paternal mean age between 30 and 35 years old (OR =1.26, 95% CI: 0.55–2.86, P=0.59) and greater than 35 years old (OR =1.23, 95% CI: 0.75–2.02, P=0.41). Moreover, no significant differences in pregnancy rates between TESA and PESA were observed among maternal subgroups stratified by mean age (Figure S2).
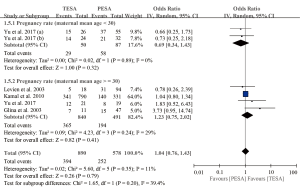
Miscarriage
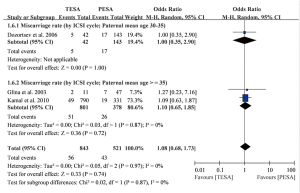
Only two studies involving 243 men receiving 263 ICSI cycles reported miscarriage rates (6,18), and pooled estimates indicated no significant difference between TESA and PESA (OR =1.08, 95% CI: 0.68–1.73, P=0.74) (Figure S3).
Publication bias
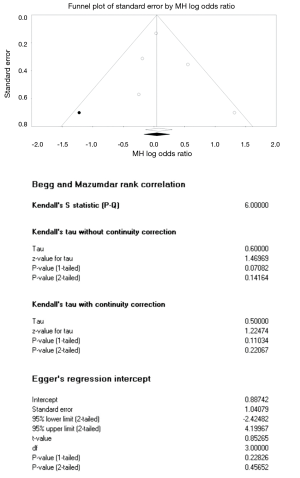
Pregnancy rate was the target outcome for publication bias testing as it was reported most frequently. The funnel plot showed no substantial asymmetry (Figure S4); moreover, Begg and Mazumdar rank correlation (tau =0.50, z-value =1.22, P=0.22) and Egger’s test (intercept =0.89, t-value =0.85, P=0.46) revealed no evidence of publication bias.
Discussion
The present synthesis identified eight studies comparing ICSI outcomes between TESA and PESA for men with OA. Reported outcomes included fertilization rate, implantation rate, pregnancy, and miscarriage rate. There were no significant differences in pregnancy and miscarriage rates between TESA and PESA treatment groups, although fertilization rate was slightly higher using PESA and implantation rate numerically higher using TESA. Thus, the choice of TESA or PESA for men with OA does not appear to influence pregnancy or miscarriage rate after ICSI, although each approach has unique advantages and disadvantages for fertilization and implantation.
In addition to yielding similar pregnancy rates, TESA and PESA share several advantages, including simplicity of the procedure. However, TESA uses a 22-G butterfly needle to penetrate into the testis with suction for fluid aspiration, and it is easier to retrieve sperm as TESA targets the main source. This ease of access may also influence quality and quantity. The quality of sperm may differ among reproductive regions in older males. In particular, the quality and quantity of sperm from the distal end of the testis may be inferior to sperm in the testis. This may account for the higher implantation rate using TESA compared to PESA. Furthermore, Color Doppler ultrasonography is an effective support tool for TESA guidance to avoid blood vessels and reduce hematoma formation (22). However, TESA causes testicular damage due to multiple needle penetrations, and may lead to intra-testicular hemorrhage. Other disadvantages of TESA include tunical vessel puncture and hematocele formation (23).
In contrast, PESA is usually performed using a 26-G needle to puncture directly into the epididymal ductule with suction force. The syringe is then rotated and advanced in a different direction with suction to extract fluid from the epididymis. This procedure may be repeated several times as PESA is conducted blind and depends on palpation by the surgeon to find the epididymis. Alternatively, PESA is an easy approach to repeat the sperm retrieval procedure and has been confirmed as highly effect for ICSI treatment of OA regardless of etiology (24). Sperm retrieval appears to be more effective in men with OA than testicular failure as evidenced by higher pregnancy rate and live birth rate (25). The main disadvantage of PESA is that the puncture site cannot be precisely controlled, and so the needle may miss the sperm-containing ductile. In addition, hematoma due blood vessel puncture and contamination of the sample with red blood cells have been observed (26).
Several studies have reported higher incidences of developmental impairment and congenital diseases due to DNA abnormities in children born through ICSI, whether sperm was retrieved from the testicle or epididymis (27). There is also evidence that mitochondrial DNA (mtDNA) and nuclear DNA (nDNA) are of better quality in testicular sperm than epididymal sperm (28), which may provide another explanation for the higher implantation rate using TESA. This in turn implies that PESA should be accompanied by longer-term follow-up. Open epididymal sperm aspiration is an alternative to PESA in which the puncture site is better controlled (29). However, we did not find evidence indicating that children born through TESA-ICSI demonstrate better development during long-term follow-up than those born through PESA-ICSI.
This meta-analysis supports the conclusion of a previous synthesis in the Cochrane Database that there is little evidence for the superiority of TESA or PESA (30). Our synthesis revealed compatible outcomes using TESA or PESA, although TESA yielded higher implantation rates than PESA in men older than 35 years. A previous meta-analysis also found a trend showing that paternal age may be a factor affecting ICSI using different retrieval techniques, although that synthesis focused on cryptozoospermia (14). On the contrary, our synthesis revealed a lower fertilization rate using TESA compared to PESA. Therefore, the present evidence cannot provide guidance for choosing TESA over PESA or vice versa for ICSI among men with OA. As these two sperm retrieval techniques have similar efficacy, we suggest that future studies should focus instead on safety outcomes. Clinicians may then be better able to choose the superior technique based on benefit-risk assessment.
There are some limitations of our meta-analysis. First, we did not compare efficacy for future pregnancies or miscarriage rate following PESA or TESA failure. Both procedures can damage tissue, and there is no guarantee of success even though sperm aspiration is a route technique for artificial reproduction. Thus, OA patients may receive either PESA or TESA multiple times. In this light, it may also be valuable to compare PESA to TESA for cost and post-operation pain. Second, our quantitative syntheses are based on only eight studies (at most) because we rejected those reporting percentages without the denominator. Moreover, some outcome analyzes were based on only two or three studies, although there was no serious bias in these cases. Third, some of the included studies did not report maternal age, an important factor influencing ICSI success. Therefore, we could not examine how maternal age affects pooled implantation and miscarriage rates following PESA and TESA. Another potential problem is that two studies were from the same institute in Brazil and had a partially overlapping in study period. However, they reported different outcomes, and no data of them can be pooled together. Therefore, our results were not biased by overlapping cohorts.
Conclusions
The pooled evidence in this meta-analysis confirms that TESA and PESA have similar efficacy for men with OA, with both yielding implantation rates higher than 80%. On the other hand, the two approaches may have different risks in clinical practice. TESA is the bigger target for sperm aspiration but may be more harmful than PESA. Alternatively, while PESA is less damaging, it may have to be performed several times as the epididymal ductule is much smaller than the testis. Thus, doctors should base the decision on whether to use TESA or PESA for sperm retrieval according to accessibility and risk of damage.
Supplementary
Supplementary file 1 Data source and search strategy
Primary search strategy
1. Obstructive azoospermia;
2. OA;
3. #1 OR #2;
4. Intracytoplasmic sperm injection;
5. ICSI;
6. Vitro fertilization;
7. IVF;
8. #4 OR #5 OR #6 OR #7;
9. #3 AND #8.
Embase
('obstructive azoospermia'/exp OR 'obstructive azoospermia' OR oa) AND ('intracytoplasmic sperm injection'/exp OR 'icsi' OR 'injection, intracytoplasmic sperm' OR 'intracytoplasmic sperm injection' OR 'sperm injections, intracytoplasmic' OR 'vitro fertilization' OR 'ivf'/exp OR ivf)
PubMed
((obstructive azoospermia OR OA) AND (((intracytoplasmic sperm injection OR ICSI) OR vitro fertilization) OR IVF))
ScienceDirect
(obstructive azoospermia OR OA) AND (intracytoplasmic sperm injection OR ICSI OR vitro fertilization OR IVF)
Web of Science
Topic: (obstructive azoospermia OR OA) AND Topic: (intracytoplasmic sperm injection OR ICSI OR vitro fertilization OR IVF)
Acknowledgments
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
References
- Practice Committee of American Society for Reproductive Medicine in collaboration with Society for Male Reproduction and Urology. The management of infertility due to obstructive azoospermia. Fertil Steril 2008;90:S121-4. [Crossref] [PubMed]
- Palermo G, Joris H, Devroey P, et al. Pregnancies after intracytoplasmic injection of single spermatozoon into an oocyte. Lancet 1992;340:17-8. [Crossref] [PubMed]
- Palermo GD, Schlegel PN, Hariprashad JJ, et al. Fertilization and pregnancy outcome with intracytoplasmic sperm injection for azoospermic men. Hum Reprod 1999;14:741-8. [Crossref] [PubMed]
- Borges E Jr, Rossi-Ferragut LM, Pasqualotto FF, et al. Testicular sperm results in elevated miscarriage rates compared to epididymal sperm in azoospermic patients. Sao Paulo Med J 2002;120:122-6. [Crossref] [PubMed]
- Kamal A, Fahmy I, Mansour R, et al. Does the outcome of ICSI in cases of obstructive azoospermia depend on the origin of the retrieved spermatozoa or the cause of obstruction? A comparative analysis. Fertil Steril 2010;94:2135-40. [Crossref] [PubMed]
- Dozortsev D, Neme R, Diamond MP, et al. Embryos generated using testicular spermatozoa have higher developmental potential than those obtained using epididymal spermatozoa in men with obstructive azoospermia. Fertil Steril 2006;86:606-11. [Crossref] [PubMed]
- Ezeh UI. Beyond the clinical classification of azoospermia: opinion. Hum Reprod 2000;15:2356-9. [Crossref] [PubMed]
- Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 2009;6:e1000097. [Crossref] [PubMed]
- Chen LS, Chen WC, Kang YN, et al. Effects of transabdominal preperitoneal and totally extraperitoneal inguinal hernia repair: an update systematic review and meta-analysis of randomized controlled trials. Surg Endosc 2019;33:418-28. [Crossref] [PubMed]
- Lin EY, Kuo YK, Kang YN. Effects of three common lumbar interbody fusion procedures for degenerative disc disease: A network meta-analysis of prospective studies. Int J Surg 2018;60:224-30. [Crossref] [PubMed]
- Lin TM, Chi JE, Chang CC, et al. Do etoricoxib and indometacin have similar effects and safety for gouty arthritis? A meta-analysis of randomized controlled trials. J Pain Res 2018;12:83-91. [Crossref] [PubMed]
- Higgins JP, Green S. Cochrane handbook for systematic reviews of interventions. 2008. Available online: https://handbook-5-1.cochrane.org/
- Yu Y, Wang Q, Zhang H, et al. Relationship of paternal age with outcome of percutaneous epididymal sperm aspiration and testicular sperm aspiration: Intracytoplasmic sperm injection with obstructive azoospermia. Int J Clin Exp Med 2017;10:5338-43.
- Ku FY, Wu CC, Hsiao YW, et al. Association of sperm source with miscarriage and take-home baby after ICSI in cryptozoospermia: a meta-analysis of testicular and ejaculated sperm. Andrology 2018;6:882-9. [Crossref] [PubMed]
- Seftel A.. Does the outcome of ICSI in cases of obstructive azoospermia depend on the origin of the retrieved spermatozoa or the cause of obstruction? A comparative analysis: Editorial comment. J Urol 2011;186:242-3. [Crossref]
- Hammoud I, Albert M, Bailly M, et al. Is testicular sperm more efficient than epididymal sperm for ICSI in patients with obstructive azoospermia? Fertil Steril 2011;96:S52. [Crossref]
- Borges E Jr, de Almeida Ferreira Braga DP, de Sousa Bonetti TC, et al. Artificial oocyte activation with calcium ionophore A23187 in intracytoplasmic sperm injection cycles using surgically retrieved spermatozoa. Fertil Steril 2009;92:131-6. [Crossref] [PubMed]
- Glina S, Fragoso JB, Martins FG, et al. Percutaneous epididymal sperm aspiration (PESA) in men with obstructive azoospermia. Int Braz J Urol 2003;29:141-5; discussion 145-6. [Crossref] [PubMed]
- Levine LA, Dimitriou RJ, Fakouri B. Testicular and epididymal percutaneous sperm aspiration in men with either obstructive or nonobstructive azoospermia. Urology 2003;62:328-32. [Crossref] [PubMed]
- Semião-Francisco L, Braga DP, Figueira Rde C, et al. Assisted reproductive technology outcomes in azoospermic men: 10 years of experience with surgical sperm retrieval. Aging Male 2010;13:44-50. [Crossref] [PubMed]
- Wang S, Li X, Wu X, et al. The study of microtraumatic sperm retrieval and intracytoplasmic sperm injection for the obstructive azoospermia. Chinese Journal of Andrology 2006;20:29-31.
- Belenky A, Avrech OM, Bachar GN, et al. Ultrasound-guided testicular sperm aspiration in azoospermic patients: a new sperm retrieval method for intracytoplasmic sperm injection. J Clin Ultrasound 2001;29:339-43. [Crossref] [PubMed]
- Shah R.. Surgical sperm retrieval: Techniques and their indications. Indian J Urol 2011;27:102-9. [Crossref] [PubMed]
- Esteves SC, Lee W, Benjamin DJ, et al. Reproductive potential of men with obstructive azoospermia undergoing percutaneous sperm retrieval and intracytoplasmic sperm injection according to the cause of obstruction. J Urol 2013;189:232-7. [Crossref] [PubMed]
- Esteves SC, Prudencio C, Seol B, et al. Comparison of sperm retrieval and reproductive outcome in azoospermic men with testicular failure and obstructive azoospermia treated for infertility. Asian J Androl 2014;16:602-6. [Crossref] [PubMed]
- Shrivastav P, Nadkarni P, Wensvoort S, et al. Percutaneous epididymal sperm aspiration for obstructive azoospermia. Hum Reprod 1994;9:2058-61. [Crossref] [PubMed]
- Fischer R, Baukloh V, Naether OGJ, et al. Andrology: CASE REPORT Pregnancy after intracytoplasmic sperm injection of spermatozoa extracted from frozen-thawed testicular biopsy. Hum Reprod 1996;11:2197-9. [Crossref] [PubMed]
- O'Connell M, McClure N, Lewis SE. Mitochondrial DNA deletions and nuclear DNA fragmentation in testicular and epididymal human sperm. Hum Reprod 2002;17:1565-70. [Crossref] [PubMed]
- Blok JM, van Roekel C, Oude Ophuis RJA, et al. Open epididymal spermatozoa aspiration for obstructive azoospermia. Andrologia 2019;51:e13218. [Crossref] [PubMed]
- Van Peperstraten A, Proctor ML, Johnson NP, et al. Techniques for surgical retrieval of sperm prior to intra-cytoplasmic sperm injection (ICSI) for azoospermia. Cochrane Database Syst Rev 2008.CD002807. [PubMed]